Sydney Choi – Year 3 (Student)
Abisola Kehinde (Mentor)
Introduction
In Canada, 90% of diabetes cases are categorized into type 2, a condition that occurs due to the improper regulation and use of glucose (Public Health Agency of Canada, 2022). The Public Health Agency of Canada (2022) also states that insulin, produced by the liver, is the regulating hormone for glucose and type 2 diabetes can be a result of the malfunction of insulin and therefore cannot regulate blood glucose effectively. This can lead to conditions such as hyperglycemia which is more commonly known as high blood sugar (Mayo Clinic, 2022). While the Canadian Diabetes Association (n.d.) outlines some ways to manage this condition such as by “eating healthy meals and snacks, enjoying regular physical activity and monitoring your blood sugar with a home blood glucose meter”, the use of an organic compound to help regulate type 2 diabetes may be a way to maximize efficiency and accessibility. Current medications for type 2 diabetes include metformin, SGLT2 inhibitors, GLP-1 receptor agonists, DPP-4 inhibitors, sulfonylureas, thiazolidinediones, insulin, and others, each working through different mechanisms to lower blood glucose levels and often chosen based on patient-specific factors like cardiovascular risk, kidney function, and weight (Mayo Clinic Staff, 2025). However, these medications are quite expensive both to make and obtain, therefore, the accessibility to these medications are highly reduced (Taylor, 2020). The use of a common organic compound could increase the accessibility of developing treatments for illnesses related to glucose regulation.
α-Amylase is an enzyme that plays a key role in digesting carbohydrates, particularly starches (Encyclopaedia Britannica, 2024). It contributes to the function of glucose release from the pancreas. High levels of α-amylase may be an indication of a condition that affects the pancreas or salivary glands (Akinfemiwa et al., 2023). As the protein is an enzyme required to break down starches into glucose for the body to absorb, it plays a factor in hyperglycemia and type 2 diabetes (Mayo Clinic 2022). Amylase inhibitors are tools used to inhibit the function of amylase in the body (Beloit College, 2024). These substances are able to prevent the process of breaking down starches into glucose. Therefore, the inhibitors are known to “prevent diseases such as diabetes, hyperglycemia, hyperlipidemia, and obesity” (Gong et al., 2020).
Studies have shown that polyphenols are effective α-amylase and α-glucosidase inhibitors (Kim et al., 2016). Polyphenols are organic compounds that are characterized by containing more than one phenolic hydroxyl group (Kim et al., 2016). Though they are not essential nutrients, they have been shown to have many health benefits such as protection against development of certain cancers, cardiovascular diseases and activating the immune system to fight off diseases (Nock, 2021; WebMD, 2022). Additionally, polyphenols are abundant in plants including a variety of fruits, black currants, dark chocolate and hazelnuts which are often highly accessible foods that are already integrated into daily diet (UCLA Health, 2022). In a 2021 study, young apple polyphenols and other phenolic compounds were tested for inhibitory activities against porcine alpha-amylase (Plamada & Vodnar, 2021). It concluded that it is possible for starch digestion by α-amylase to be slowed and therefore controlled by polyphenols’ ability to inhibit the function of the enzyme (Plamada & Vodnar, 2021).
There has been past research on extracting polyphenols from fruits. A study conducted by Annegowda et al. (2010) show that effective methods of extraction include sonication to extract polyphenols from Terminalia catappa L plant. Additionally, using 50% and 70% EtOH as a solvents along with sonication is found to yield extracts rich in phenolic and flavonoid content (Dirar et al., 2018).
This study will aim to further investigate the relationship between α-amylase and polyphenols and how the compounds can inhibit the activity of the enzyme. Using polyphenolic extract from strawberries, an accessible fruit, the inhibition of starch breakdown will be observed through a starch-iodine reaction. A difference in colour of the solution will indicate that the polyphenol has successfully inhibited the α-amylase enzyme. Investigating the ability of polyphenols in strawberries to inhibit enzyme activity may help increase the accessibility of treatments for illnesses such as type 2 diabetes as it would improve the ability to control glucose levels in the body using commonly available fruits.
Materials and Methods
Preparation of Fruit for Extraction
The fruit used for this extraction were fresh strawberries (Driscoll’s). They were purchased 1-2 days prior to the extraction and held in a refrigerator at 3ºC before being washed and thoroughly dried. Then, the stems were removed and the flesh was cut into small pieces before being crushed. The strawberries were weighed to be 100g before the addition of any chemicals.
Extraction
500mL of methanol (Fisher Scientific) was left in the freezer for 48 hrs at -18ºC before it was added to the strawberries. The ratio of strawberries to methanol is 1:5. The solution was left in the ultrasonic bath (Creworks) at room temperature (21ºC) for one hour, contained in a beaker covered by aluminum foil. After sonication, the solution was centrifuged for 2 minutes at 4000 rpm in a centrifuge (Spectrafuge) to separate the solution from remaining strawberry flesh.
Starch Substrate for Enzyme Inhibition
To 50mL or distilled water in a 500mL beaker, 2.0g of Yupik Organic Soluble Potato Starch was dissolved. The beaker was then placed on a hotplate (Salton) set to 200ºC and constantly stirred until the white, opaque liquid started to bubble and the solution turned into a clear, homogeneous gelatinous slurry. The hotplate was then turned off and the beaker was set aside to cool to room temperature. Once the solution had cooled, 100mL of distilled water was added and stirred to combine.
Enzyme Inhibition
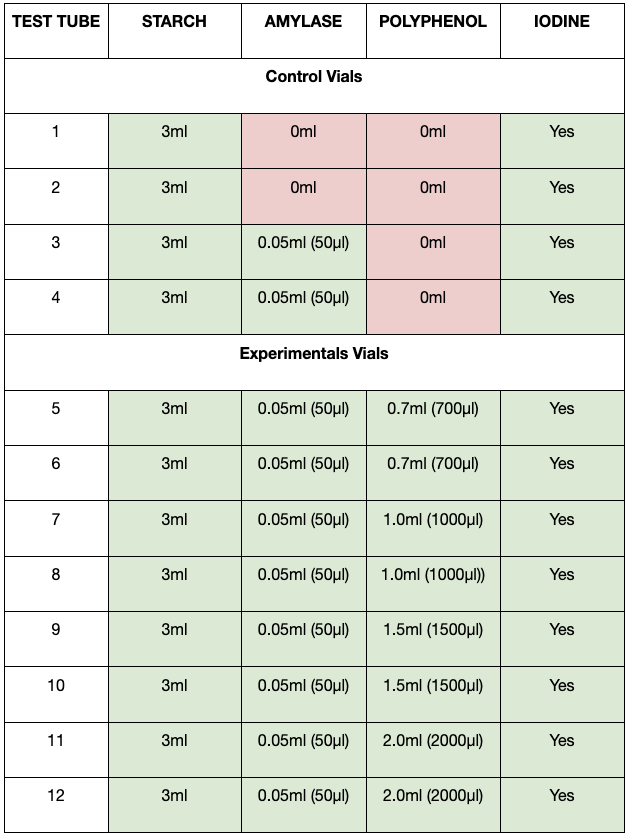
To 20ml of distilled water, 1.5g of amylase enzyme powder (LD Carlson) was dissolved to form an amylase enzyme solution. To test the enzyme inhibiting function of the polyphenol, twelve 14ml plastic vials (Ambion) were labelled from 1-12 and 3mL of the starch solution was added to each vial using a 3mL transfer pipette.Vials 1-4 served as controls where vials 1-2 contained only starch and vials 3-4 contained starch and 50μl amylase enzyme solution. Vials 3-4 were observed as a control to ensure that the amylase did, in fact, break down the starch. The remaining test tubes contained 700μl, 1000μl, 1500μl or 2000μl of the polyphenol extract using a micropipette with duplicates of each (Table 1). Two drops of iodine reagent (ALDON) were added to all test tubes (vials 1-12) after 35 minutes. The colour of the solution upon addition of the iodine indicated the presence of starch. A dark blue or indigo would indicate presence of starch while a clear, pale yellow would indicate an absence of starch. The experiment was replicated for a total of three trials.
Table 1. Table listing quantity of contents in each test tube.
Results
Overall, the three trials yielded clear and relatively consistent results . The control variable that consisted of just starch was blue upon addition of the iodine across all three trials. The control variable containing starch and amylase remained a clear yellow colour upon the addition of iodine which indicates that there is no starch present. However, in Vial 4 of Trial 1, the colour of the solution was not a clear yellow colour but a dark blue colour (Figure 1). Trial 3 yielded clear, expected results.
All of the vials containing polyphenol extract remained a dark blue colour indicating that starch was present in the solution despite the amylase also being present. However, vials 7-12 across all three trials showed some inconsistencies as some of the vials were dark blue while others were a clearer colour. For example, although vials 9 and 10 contained the same amount of amylase, polyphenol extract, iodine and starch, they showed contrasting results as vial 9 was a clearer and lighter purple colour while vial 10 was the expected dark bluish-purple colour. Additionally, in vial 12 of Trial 3, the colour of the solution was slightly lighter and it was clearer than its duplicate, vial 11 (Figure 2).
Figure 1. Results from Trial 1
Figure 2. Results from Trial 3
Discussion
Overall, the experiment yielded relatively consistent results that suggest that the polyphenolic extract is able to inhibit the enzyme, amylase, from breaking down the starch. Nearly all vials containing different amounts of polyphenols remained a deep indigo upon the addition of iodine indicator, indicating the presence of starch even after the addition of amylase. Some of the vials that did not remain blue can be attributed to a source of error as the duplicate vials did not yield the same result. A source of error may be the size of the iodine drops as only a few were added, but the size of each drop slightly varied. Since control vials 3 and 4 demonstrated that the amylase did, in fact, break down starch, it can be concluded that the addition of the polyphenol extract caused the enzyme function to be inhibited. However, there were some inconsistencies in results such as the difference between duplicate vials. Since there was no significant colour difference between the vials containing different amounts of polyphenol, there is not a clear conclusion about how the volume of polyphenol extract affects the enzyme inhibition. This may be due to slight differences in the amount of iodine added as more iodine seems to yield a more vibrant colour, as seen in Figure 2 – vials 11 and 12, (although the measurements were made as precisely as possible) or the amount of time in between the addition of certain solutions.
Moving forwards, further areas of study could include exploring the extraction process of the polyphenols deeper. Due to limited resources, it was not possible to guarantee the presence of polyphenols in the extraction. If the experiment were to be conducted again with a greater range of resources, a spectrophotometer could be used alongside a gallic acid calibration curve to confirm the presence of polyphenols (Dirar et al., 2018). Additionally, polyphenols can be extracted from different fruits such as pomegranate and blueberries as they are both known to contain high concentrations of the compound. This experiment suggests that polyphenolic extract from strawberries were effective at inhibiting the enzyme α-amylase from breaking down starch. The findings of this experiment further demonstrate that polyphenols extracted from common fruits, can be used for amylase inhibition, and could be applied to the development of type 2 diabetes treatments, increasing the accessibility of medication for this disease.
References
Anderson, G. H., & Moore, S. E. (2016). Dietary proteins in the regulation of food intake and body weight in humans. Nutrients, 8(1), 17. https://doi.org/10.3390/nu8010017
Annegowda, H., Anwar, L., Mordi, M., Ramanathan, S., & Mansor, S. (2010). Influence of sonication on the phenolic content and antioxidant activity of Terminalia catappa L. leaves. Pharmacognosy Research, 2(6), 368. https://doi.org/10.4103/0974-8490.75457
Balfour, J. A., & Buckley, M. M. (1991). Alpha-amylase and human digestion. Frontiers in Nutrition, 15(3), 1-10. https://doi.org/10.1016/PMC6273793
Beloit College. Amylase activity: Enzyme kinetics lab. Retrieved October 7, 2024, from https://chem.beloit.edu/classes/Chem260/Labs/Amylase/Amylase%20Activity.pdf
Canadian Diabetes Association. (n.d.). Type 2 Treatment. Diabetes Canada. https://www.diabetes.ca/about-diabetes/type-2/treatment
Dirar, A., Alsaadi, D., Wada, M., Mohamed, M., Watanabe, T., & Devkota, H. (2018). Effects of extraction solvents on total phenolic and flavonoid contents and biological activities of extracts from Sudanese medicinal plants. South African Journal of Botany, 120, 261–267. https://doi.org/10.1016/j.sajb.2018.07.003
Encyclopaedia Britannica. (2024). Alpha-amylase. In Britannica. Retrieved October 7, 2024, from https://www.britannica.com/science/alpha-amylase
Gong, L., Feng, D., Wang, T., Ren, Y., Liu, Y., & Wang, J. (2020). Inhibitors of α-amylase and α-glucosidase: Potential linkage for whole cereal foods on prevention of hyperglycemia. Food Science & Nutrition, 8(12), 6320–6337. https://doi.org/10.1002/fsn3.1987
Kim, Y., Keogh, J. B., & Clifton, P. M. (2016). Polyphenols and Glycemic Control. Nutrients, 8(1), 17. https://doi.org/10.3390/nu8010017
Mayo Clinic. (2023, July 7). Hyperglycemia: Symptoms and causes. https://www.mayoclinic.org/diseases-conditions/hyperglycemia/symptoms-causes/syc-20373631
Mayo Clinic Staff. (2025, February 27). Type 2 Diabetes – Diagnosis and treatment. Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/type-2-diabetes/diagnosis-treatment/drc-20351199
Plamada, D., & Vodnar, D. C. (2021). Polyphenols-Gut Microbiota Interrelationship: A Transition to a New Generation of Prebiotics. Nutrients, 14(1), 137. https://doi.org/10.3390/nu14010137
Public Health Agency of Canada. (2022, December 2). Framework for diabetes in Canada. Canada.ca. https://www.canada.ca/en/public-health/services/publications/diseases-conditions/framework-diabetes-canada.html
Siemens Stiftung. (2017, October 11). Enzymes: Amylase and starch digestion [Video]. YouTube. https://www.youtube.com/watch?v=nHGPoSTYW50
Taylor S. I. (2020). The High Cost of Diabetes Drugs: Disparate Impact on the Most Vulnerable Patients. Diabetes care, 43(10), 2330–2332. https://doi.org/10.2337/dci20-0039
UCLA Health. (2023, June 12). Polyphenols can be found in many fresh foods. https://www.uclahealth.org/news/article/polyphenols-can-be-found-in-many-fresh-foods#:~:text=Fortunately%20for%20anyone%20who%20wants,%2C%20coffee%2C%20hazelnuts%20and%20pecans
WebMD. Foods high in polyphenols. Retrieved October 7, 2024, from https://www.webmd.com/diet/foods-high-in-polyphenols