Caryl Szeto – Life Science, Year 2
Abstract
Plastic is used throughout the world; however, its growing presence in our environments and our own bodies is cause for concern. It’s been found in Antarctica, the Arctic, Mount Everest, and even the Mariana Trench (Li et al., 2023). With new research coming to light regarding the effects of microplastics (MPs) on human health, and their growing existence within the human body, it is essential that people understand whether their water is contaminated, and where the contamination may be originating from. This study explores different methods of testing for MPs in water using relatively readily available household materials. One of the methods involved creating hard water by adding calcium carbonate to water, before boiling it to allow the calcium carbonate to bind to MP’s (Ries, 2024). The second method used olive oil to bind to MP’s in water by being shaken with water in a closed bottle (Scopetani et al., 2020). The boil water method yielded 3.51g of precipitate for the MP sample, compared to 2.40g for the distilled water sample. Finally, the oil separation method filtered out 16.91g of oil in the MP sample, but only 12.59g of oil in the distilled water sample. These findings suggest that both testing methods are valid tests for MPs in water. Overall, as MP testing methods become more available, they may be standardized and used to positively influence consumer behaviour and policy.
Introduction
Microplastics (MPs) are defined as “small plastic particles that come from the degradation of plastics” and do not originate naturally (Ziani et al., 2023). They are defined based on their size, as MPs have a diameter of less than 5mm. Furthermore, MP is a general term, used to describe different shapes, sizes, and types of plastics that are insoluble in water and non-degradable (Ziani et al., 2023).
MPs have been found in animals, drinking water, food and air. In fact, small concentrations found in water samples means that humans may be drinking MPs, or even inhaling them in the shower (Li et al., 2020). Furthermore, they are defined as “ubiquitous in all compartments,” as they have been found in human stool, soil, snow, rain, beach sands and deep environments (Li et al., 2020; Ziani et al., 2023). The presence of low performing waste management, such as ineffectively collecting finished plastic waste, exacerbates the MP pollution issue. This contributes to the fact that around 65 million MP particles are released into water every day after being treated by water and sewage treatment plants (Li et al., 2023).
MPs have been found to contact humans in many different ways, such as: oral intake (food, containers, etc.), inhalation (air) and skin contact (hygiene products, etc.) (Li et al., 2023). In fact, one of the biggest areas for contamination is caused due to transfer of MPs between containers and their contents, for instance, food products in plastic packaging (Ziani et al., 2023). Combined, these modes of contact cause an average person to intake 39,000-52,000 MP particles, every year (Li et al., 2023). This is important to note, as researchers have found MPs in blood, the spleen, liver, colon, placenta and breast. The presence of MPs in one’s body can lead to many long-term health effects, such as DNA damage, neurotoxicity, metabolic disorders, and reproductive dysfunctions (Li et al., 2023).
There are multiple ways that MPs can enter water systems and pollute the environment. A Material Flow Analysis done in Canada, showed that 60,100 tons of MPs were released into Canada’s environment in 2016 (Sherlock, 2023). The three highest sources of MPs were from tires, paint fragments, and microfibers. The areas where this happened most often were roadsides due to emissions, direct release to soils, and direct release to surface water, with roadsides and surface water receiving the most MPs (Sherlock, 2023). In fact, MPs have been found in rainwater and storm water, which allows them to be exchanged in different environments (Wei et al., 2023). Considering how the Coquitlam watershed, which provides 2.8 million people with drinking water, is filled with rain and melting snow, the presence of MPs in rainwater may be one way they enter the Coquitlam water reservoir (“Watersheds and Reservoirs ,” n.d.).
Considering the prevalence of MPs and the impact they have on human health, the need for proper testing of MPs within households is essential, so that people can understand whether there are MP’s present in their water. There are two methods of testing for MPs in water that have been identified as cost effective and adaptable for household use. Although the University of British Columbia is currently working on a water testing kit, it has not reached the market yet, and may not be accessible to those outside of the province or country (Griffiths, 2024). This is why is it essential for different testing techniques to be developed.
Within the context of Vancouver, the first water testing technique to be considered is the boil water method. Researchers in China discovered that boiling water removed up to 90% of MPs in water (Ries, 2024). Calcium carbonate in the hard water would encapsulate MPs, making them easy to filter out. Because water in Vancouver is considered soft, there are less minerals present in the water (Metro Vancouver, n.d.). Consequently, it is assumed that calcium carbonate would need to be added to Vancouver water to replicate these results. By weighing the filter paper before and after filtering, the amount of MPs captured should, in theory, be revealed.
The second water testing technique is the oil separation method. Researchers in Europe found that adding oil to sludge containing MPs allowed for easier filtration (Lekše et al., 2024). MPs are considered oleophilic, meaning they are attracted to oil (Scopetani et al., 2020). This may be because both MPs and oil are non-polar (Venture Media, 2021). By shaking a sample of water with olive oil, to fully mix it, and then allowing the oil and water to separate, MPs should be transferred to the olive oil. Then, by filtering out the oil and dissolving it with alcohol, the amount of MPs filtered out should be revealed.
Overall, by comparing these two testing techniques and considering their potential for use within households, we may find more cost-effective and universal ways of testing for MPs in water. Furthermore, in the future, these testing techniques could be used on rainwater or to test water filters, which would give insight on the effectiveness of water filters and local relevance of MPs in the Vancouver area. This would allow for general global research to be applied at a local level.
MP’s have been found throughout the world and throughout our living spaces. From waterways, to drinking water, to the human body, they are present everywhere in our everyday lives. Due to their impact on human health, is it essential that people are aware of the quality of their water, and where the contamination may be stemming from.
Materials and Methods
Water samples
For both the boil water method and olive oil separation method, the same solution of MP contaminated water was used as a positive control. According to Akbulut et al. (2024), the highest number of MPs released into a beverage sample was when 50°C fluid soaked in a plastic lined cup for 20 minutes. In order to ensure proper saturation of MPs in the plastic water solution, 400ml of boiling tap water was soaked in a plastic-lined paper cup for 20 minutes. Furthermore, distilled water was used as a negative control in this experiment because water distillers will “separate microplastics from water during the distillation process” (Woodard, 2023). Finally, in order to test the ability of Brita water filters to filter out MP’s as they advertise, Vancouver tap water filtered through a Brita standard filter was used as a sample (Brita Hub, n.d.). This was based on the assumption that MPs would be present in Vancouver tap water. After preparing these samples, each group went through testing as described below.
Boil Water Method
According to HydroFLOW USA. (2022), 3g of calcium carbonate, 6g of calcium hydroxide, 3g of magnesium sulfate, and 3g of calcium chloride should be added to 2000ml of water to increase water hardness to +3000ppm. Combining this research with an article by Ries. (2024), 2g of calcium carbonate (Heiltropfen), was added to 200ml of tap water. This ensured proper saturation of the water. This solution was used as the hard water sample, that was mixed with the positive or negative control.
Based on the methodology from Ries (2024), 200ml of the plastic water solution was added to 200ml of the calcium carbonate sample. The solution was then boiled for 5 minutes and left untouched for 10 minutes. Two sheets of layered coffee filter paper (Compliments) were used to filter out the calcium carbonate precipitate (Figure 1). The two filter papers were weighed twice, once before filtering the precipitate, and once after drying on a hot plate. This allowed for the difference in weight to be calculated, and in theory, also the weight of the MP’s captured in the calcium carbonate precipitate. This same method above was repeated for the negative control, and sample of filtered water.
Figure 1: Precipitate formation filtered through coffee filter paper and then dried.
Oil Separation Method
400ml of the plastic sample was mixed with 20ml of extra virgin olive oil into a plastic bottle with a lid. The bottle was shaken by hand for 1 minute and 30 seconds before resting for 15 minutes (Scopetani et al., 2020). The top layer of oil was extracted with a spoon and pipette, and was placed on a layer of two coffee filter papers (Figure 2). Any remaining water was filtered through the paper while the oil remained. The filter papers were weighed before and after the separation process, and this method was repeated for all other samples. The weight of the oil captured through the filter papers should represent the amount of MP’s that are bound to the oil.
Figure 2: Oil and water separation through filter paper.
Results
Boil Water method
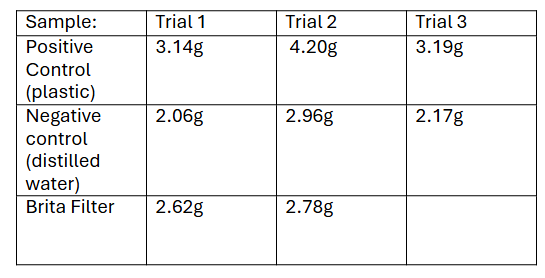
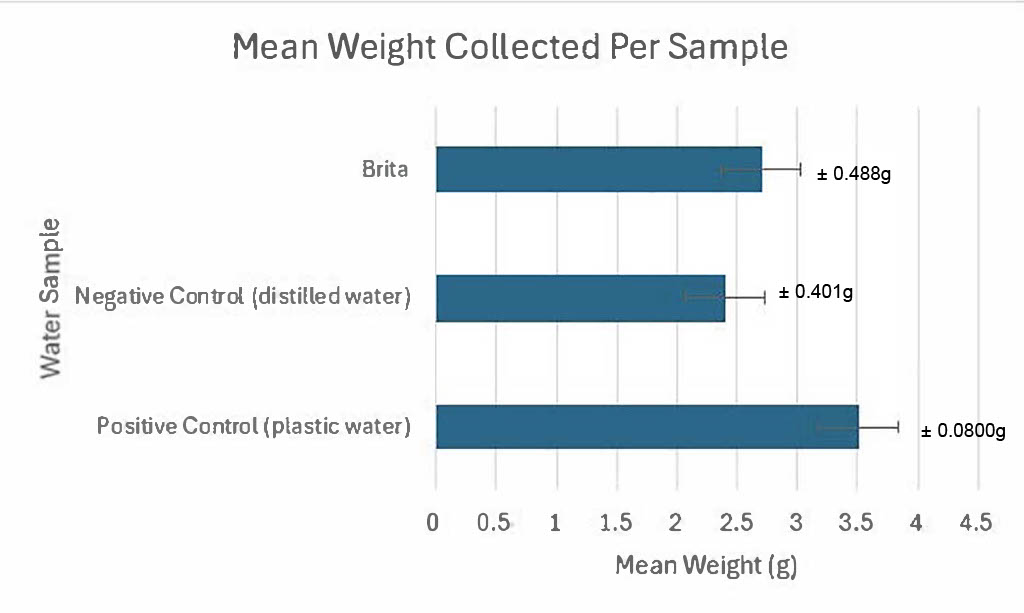
The boil water method yielded results where the plastic water sample yielded more calcium carbonate, compared to the distilled and Brita water samples (Table 1). Results for the positive control had a mean weight of 3.51g ± 0.488g. The negative control had a mean weight of 2.40g ± 0.401g. Finally, the Brita filter sample had a mean weight of 2.70 g ± 0.0800g (Figure 3).
Table 1: Weight difference from the filter papers containing calcium carbonate precipitate after being boiled with water samples.
Figure 3: Mean weight collected per water sample
Oil Separation Method
The oil separation method yielded similar results to the boil water method, whereby the plastic water sample yielded greater precipitate, compared to the distilled and Brita water samples (Table 2). Results for the positive control had a mean weight of 16.91g ± 0.690g. The negative control had a mean weight of 12.60g ± 0.455g. Finally, the Brita filter sample had a mean weight of 14.41g ± 0.455g (Figure 4).
Table 2: Oil amount captured through coffee filter paper, after being mixed through water samples.
Figure 4: Mean weight collected per water sample
Discussion
This study demonstrates the viability of accessible, household testing methods for microplastics. Both tests yielded more precipitate compared to controls, and it is an assumption that this difference in weight represents the amount of MPs captured. The results for both testing methods demonstrate that they are credible tests, as the samples with more MPs had higher differences in weight. This demonstrates how the calcium carbonate and olive oil did bind to the MPs. Furthermore, for both test methods, the precipitate weight of Brita filtered samples fell between that of the negative and positive control samples. However, it should be noted that unfiltered Vancouver tap water was not tested, meaning direct comparisons between filtered and unfiltered water can not be made. Additionally, it should be noted that precipitate formation could be due to other contaminants besides MPs.
The comparison of the two water testing methods underscores the central theme of this study – the potential for both methods to be present in the average household. Given that olive oil is more readily available than calcium carbonate, the oil separation method appears to be a more accessible option for testing household water for MPs. Furthermore, while both methods were relatively quick to implement, each requiring less than an hour to complete two trials, the oil separation method was notably faster compared to the boil water method.
These findings have similar results to other similar studies. As seen in the results from Akbulut et al. (2024), the plastic water boiled in a plastic lined cup appeared to contain more MPs, compared to distilled and filtered water. Furthermore, in Ries (2024), calcium carbonate precipitate formed around MPs, which was also demonstrated in weight differences from Table 1. The method of manually adding calcium carbonate to water to mimic hard water seems to be effective, due to these similar results. Finally, olive oil appears to have bound to MPs in the results from the present study, as well as the results from Lekše et al. (2024). On the other hand, Lekše et al., mainly explored the size and type of MP’s captured, whereas this study focused on the weight differences of oil containing MPs. Olive oil seems to be an effective solution that can bind to MPs, as although this study tested water rather than plastic sludge, similar results were yielded.
In the future, these tests can be introduced to the Vancouver public as possible methods for testing for MPs in households. Municipalities could implement them during water quality crises as simple yet effective ways of testing for MPs, and there is a potential for these testing methods to become standardized. The implication of this could be a larger emphasis on the presence of MPs in our daily lives. Furthermore, it may reveal the high amounts of MPs found in coffee cups. Hopefully, a new alternative to plastic lining could replace coffee cups, to reduce intake of MPs. Overall, as more accessible MP testing methods are introduced to the public, consumer behaviour and policy may be affected and could drive positive change.
This study relied heavily on weighing filter paper to gather results. There were some discrepancies with the digital scaled used to weigh the papers, as although each individual paper was the same, they varied in weight by 0.1-0.5 grams. This could impede the validity of the data seen in the results section. Furthermore, a TDS meter was used to test the hardness of the water for the boil water test; however, it varied in its reading, with a range of 30-70ppm. As a result of this large uncertainty value, all quantitative data on the water hardness for these samples were dropped.
When filtering out the oil from the water, it should be dissolved using ethanol before being weighed. Unfortunately, adding ethanol to the coffee filter papers meant they would dissolve, so this method was not used for weighing. This means any water that was not filtered out fully could have affected the weight of the sample. To combat this, each filter paper sample was left to run for 3 minutes, or until no clear fluid leaked from the paper.
The original experiment planned to also test rain water, tap water and river water to determine if filters are able to remove MPs from water, and to determine where MPs in our tap water may be originating from. Testing for rainwater was not possible under time constraints for this project, but should be addressed in the future. Additionally, less tests were completed for the Brita filter samples due to time constraints, but more should be completed in the future to ensure validity of results.
Overall, findings from the present study suggest that both the oil separation method and the boil water method are effective ways of testing for MPs in water. Considering the health impacts of MPs, their wide-spread presence, and the lack of accessible water testing methods, this study may help shed light on the prevalence of MPs in everyday life. Furthermore, it may make water testing more accessible within the average household. By improving access to MP testing, this research may contribute to more informed water safety practices and greater public awareness of microplastic contamination.
References
Akbulut, S., Akman, P., Tornuk, F., & Yetim, H. (2024). Microplastic Release from Single-Use Plastic Beverage Cups. MDPI Foods.
Brita Hub. (n.d.). Brita HubTM Replacement Filter for Brita Hub. Retrieved March 12, 2025, from https://www.brita.com/products/hub-filter/
Griffiths, N. (2024). How much plastic is in your tap water? New UBC research may lead to easy home tests. Vancouver Sun. https://vancouversun.com/news/new-ubc-research-may-lead-easy-home-tests-plastic-tap-water
HydroFLOW USA. (2022, August 22). How To Make Hard Water [Video recording]. https://www.youtube.com/watch?v=_40hWeUTtcY&t=30s
Lekše, N., Žgajnar Gotvajn, A., Zupančič, M., & Griessler Bulc, T. (2024). Oil-based extraction as an efficient method for the quantification of microplastics in environmental samples. Environmental Sciences Europe, 36(1), 68. https://doi.org/10.1186/s12302-024-00898-6
Li, Y., Li, W., Jarvis, P., Zhou, W., Zhang, J., Chen, J., Tan, Q., & Tian, Y. (2020). Occurrence, removal and potential threats associated with microplastics in drinking water sources. Journal of Environmental Chemical Engineering, 8(6), 104527. https://doi.org/https://doi.org/10.1016/j.jece.2020.104527
Li, Y., Tao, L., Wang, Q., Wang, F., Li, G., & Song, M. (2023). Potential Health Impact of Microplastics: A Review of Environmental Distribution, Human Exposure, and Toxic Effects. Environment & Health, 1(4), 249–257. https://doi.org/10.1021/envhealth.3c00052
Metro Vancouver. (n.d.). Water Quality and Testing . Metro Vancouver. Retrieved March 30, 2025, from https://metrovancouver.org/services/water/water-quality-testing
Ries, J. (2024). Boiling Water May Help Remove Up to 90% of Microplastics. Healthline. https://www.healthline.com/health-news/boiling-water-may-help-remove-up-to-90-of-microplastics
Scopetani, C., Chelazzi, D., Mikola, J., Leiniö, V., Heikkinen, R., Cincinelli, A., & Pellinen, J. (2020). Olive oil-based method for the extraction, quantification and identification of microplastics in soil and compost samples. Science of The Total Environment, 733, 139338. https://doi.org/https://doi.org/10.1016/j.scitotenv.2020.139338
Sherlock, C. (2023). A Big Tiny Problem: Flows of Primary Microplastics in Canada. https://uwspace.uwaterloo.ca/items/6e2227c3-a2bb-4c4d-88c1-2b052f9f918b
Venture Media. (2021). Microplastic Pollution Solution. Issuu. https://issuu.com/travelnewsnamibia/docs/flywestair_october_issuu/s/13595100
Watersheds and Reservoirs . (n.d.). Metro Vancouver. Retrieved January 21, 2025, from https://metrovancouver.org/services/water/watersheds-reservoirs
Wei, L., Yue, Q., Chen, G., & Wang, J. (2023). Microplastics in rainwater/stormwater environments: Influencing factors, sources, transport, fate, and removal techniques. TrAC Trends in Analytical Chemistry, 165, 117147. https://doi.org/https://doi.org/10.1016/j.trac.2023.117147
Woodard, J. (2023). How to Remove Microplastics from Drinking Water. Fresh Water Systems. https://www.freshwatersystems.com/blogs/blog/how-to-remove-microplastics-from-drinking-water
Ziani, K., Ionita-Mindrican, C.-B., Mitielu, M., Neacsu, S., Negrei, C., Morosan, E., Draganescu, D., & Preda, O. (2023). Microplastics: A Real Global Threat for Environment and Food Safety: A State of the Art Review. Nutrients. https://doi.org/10.3390/nu15030617