Andy Huang – Life Science, Year 2
Abstract
Catalase plays a vital role in cellular defense by breaking down hydrogen peroxide (H₂O₂) into water and oxygen, preventing oxidative damage. In this study, I aimed to identify the optimal pH and temperature conditions that maximize catalase activity in chicken and pig liver. I hypothesized that catalase functions most efficiently at neutral to slightly basic pH and moderate temperatures. To test this, I reacted liver homogenates of both chicken and pig liver with H₂O₂ across four pH levels (4, 6, 7, 9) and three temperatures (25 °C, 35 °C, 45 °C), measuring oxygen production through pressure changes. Catalase activity increased with pH, peaking at pH 7, and rose with temperature up to 35 °C before declining at 45 °C due to heat-induced denaturation. Pig liver consistently showed higher activity than chicken liver, reflecting its greater enzyme concentration. These results confirm my hypothesis and highlight the importance of environmental conditions and species-specific factors in enzyme efficiency. Optimizing these conditions contribute by enhancing catalase applications in medicine, food processing, and biotechnology.
Introduction
Catalase is a crucial enzyme that plays a vital role in detoxifying the body by breaking down hydrogen peroxide (H₂O₂) into water and oxygen. This decomposition process is essential for protecting cells from oxidative damage, as high concentrations of H₂O₂ can lead to decreased food and water intake, as well as reduced body weight (Ransy 2020). Although H₂O₂ is produced by the body as part of its defense mechanism against pathogens, excessive levels can cause oxidative stress. This stress results from the formation of highly reactive hydroxyl radicals, which decrease metabolic rate and can lead to acute toxicity, particularly in animals such as chickens and pigs (Jomova et al., 2023).
Catalase functions as a catalyst, speeding up the breakdown of H₂O₂ in the bloodstream by lowering the activation energy required for the reaction. By providing an alternative pathway, catalase allows H₂O₂ to break down into water and oxygen molecules, reducing its harmful effects on cells. (Heck et al., 2010). This catalytic efficiency is critical, as a single catalase molecule can process up to 40 million H₂O₂ molecules per second (Robinson, 2015). The enzyme is particularly abundant in the liver, where it resides in peroxisomes—small cell compartments that help manage the breakdown of H₂O₂ produced during metabolic processes. If left unchecked, reactive oxygen species (ROS), including H₂O₂, can damage cellular components such as DNA, proteins, and lipids (Checa et al., 2020).
The structure of catalase is key to its function. It contains an iron-based heme group at its active site, which is accessible through a narrow channel, ensuring that only H₂O₂ can bind and undergo decomposition. (Domínguez et al., 2010). Catalase also works alongside other antioxidant enzymes, such as superoxide dismutase, to prevent the formation of harmful hydroxyl radicals, further protecting the body from oxidative damage (Domínguez et al., 2010).
The effectiveness of catalase is influenced by two critical factors: temperature and pH. Enzyme activity generally increases with rising temperature, as higher energy levels lead to more frequent collisions between catalase and H₂O₂ molecules. (Tijskens 2001). However, if the temperature exceeds the enzyme’s optimum, it can lead to denaturation, where the enzyme’s structure breaks down, drastically reducing its activity. Similarly, catalase operates most efficiently at a neutral to slightly alkaline pH, with peak activity occurring between pH 6 and 8, and some studies showing effectiveness up to pH 11 (Bartoszek, 2006). In acidic conditions, the enzyme’s active site undergoes changes that hinder substrate binding, while in highly alkaline conditions, alterations in the enzyme’s charge reduce its solubility and shape, rendering it inactive.
The practical applications of catalase are widespread, particularly in industries such as food preservation and wastewater treatment. For instance, catalase helps prevent browning in egg products by catalyzing the breakdown of glucose, which otherwise reacts with amino acids in the egg white to cause discoloration (Espinoza-Vergara et al., 2021). Additionally, understanding the optimal conditions for catalase activity has important implications for medical treatments. For example, patients with acatalasemia—a rare genetic condition where individuals lack sufficient catalase—are more susceptible to liver damage and oxidative stress, especially when exposed to substances like alcohol (Nandi, 2019).
Ultimately, catalase plays a vital role in protecting cells from oxidative damage by breaking down H₂O₂ into water and oxygen. Understanding the factors that affect its activity, such as temperature and pH, is essential for optimizing its function in both biological systems and industrial applications. By determining the optimal conditions for catalase, one can improve its effectiveness in processes like food preservation and medical treatments, ultimately enhancing its protective role in living organisms.
Materials and Methods
The liver was prepared by grinding into little pieces. They were placed into the mortar and pestle and added drops of water to ensure that the liver was blended until smooth. After pipetting 2 mL of 3% H2O2 into each 10 mL test tubes, one scoop of liver was transferred into the tube using the scoopula, and using the Venier Kit, the rubber stopper is used to immediately seal the tube, avoiding the escape of gases. This serves as a negative control group because of using room temperature and no addition of acids or bases. Thus, there were in total 4 pH groups (pH 4, pH 6, pH 7, and pH 9) solution, dropping 1 mL of the solution into 3 tubes. Afterwards, a water bath was prepared by heating tap water using a hot plate at the desired temperature, being 3 different temperatures (25, 35, and 45 °C), with 25 °C measured from tap water acting as a negative control. Each tube was put into the water bath for 1 minute for the solution in the test tube to reach equilibrium and attain the desired temperature.
After the Vernier kit was set up to measure pressure in Kpa over time, a magnetic stirrer was inserted into the test tube and operated at a constant speed throughout all trials. One gram of liver was transferred into the test tube using a scoopula, and the tube was immediately sealed with a rubber stopper. As the reaction proceeded and pressure increased, the maximum pressure value was recorded. Each trial lasted 90 seconds. Trials were conducted for both chicken and pig liver, with each pH level paired with each temperature condition.
Results
Qualitative Observations
At 25 °C in room temperature, bubbles formed immediately once the liver came in contacts with the H2O2, increasing in size and amount as time passed. However, after the one-minute mark, the foam gradually decreased and ascended. At 35 °C, the foam formed as bubbles in H2O2 formed in at an increased speed, leading to the fastest growth in foam. At 45 °C, while the foam increased at a rapid speed initially, it reached a specific height and decreased gradually, leading to a slightly decreased speed of the foam made than in 35 °C. Most trials saw approximately the same height of the foam created. The speed in which the foam rose for neutral and basic pH was faster than those in acidic pH.
Quantitative Observation
The average oxygen produced by chicken and pig liver are shown in Figure 1 and Table 1, and Figure 2 and Table 2 respectively.
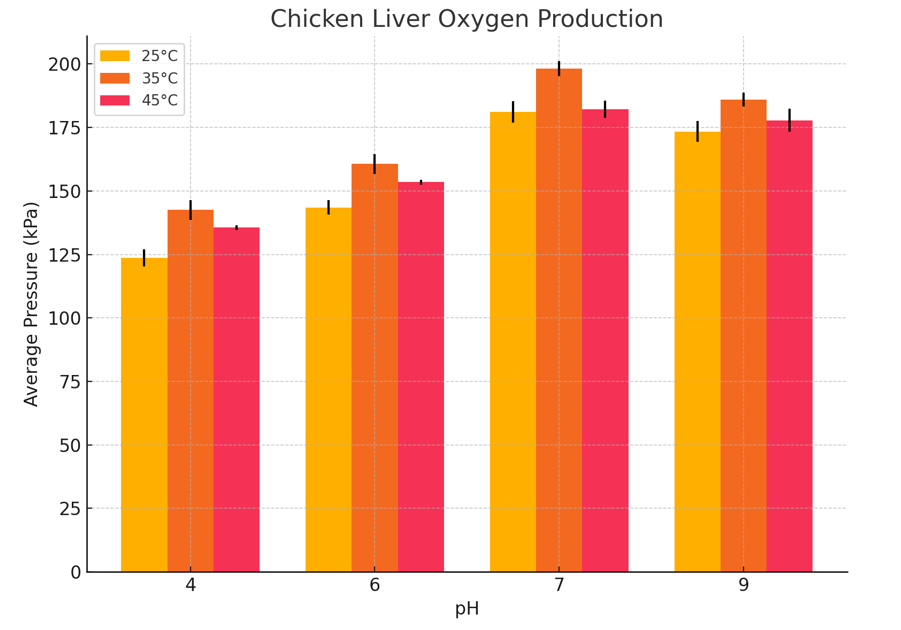
Figure 1: Production of Oxygen Measured in kPa for Chicken Liver in pH 4, 6, 7, 9 at 3 different Temperature (25°C, 35°C, 45°C)
Figure 2: Production of Oxygen Measured in kPa for Pig Liver in pH 4, 6, 7, 9 at 3 different Temperature (25°C, 35°C, 45°C)
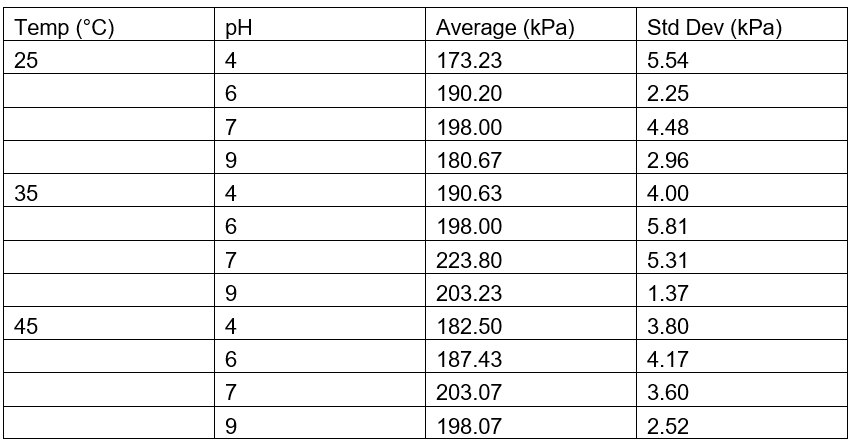
Table 1: Average Pressure for Chicken Liver Measured in kPa
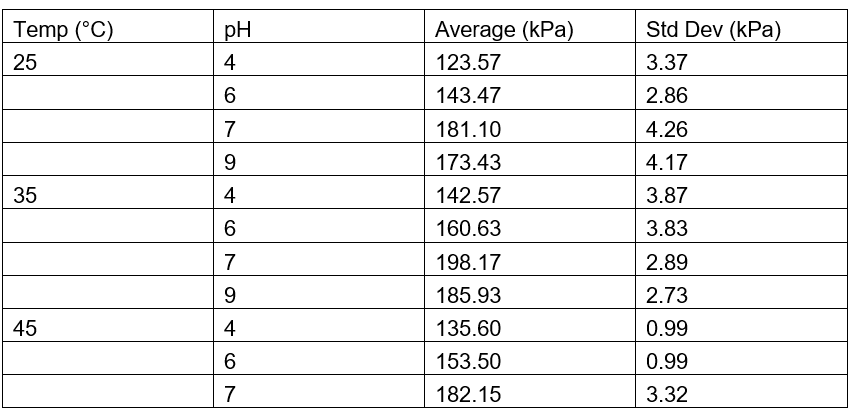
Table 2: Average Pressure for Chicken Liver Measured in kPa
Discussion
The results demonstrate a clear trend in catalase activity based on pH and temperature. Catalase activity, as measured by the maximum pressure produced from the decomposition of hydrogen peroxide (H₂O₂), increased as pH rose from acidic to neutral, peaking around pH 7 to 9, and then slightly declined at more alkaline levels. This suggests that catalase operates most efficiently at neutral to slightly basic conditions. At extreme pH values, the enzyme’s activity drops likely due to alterations in the active site structure, which hinder substrate binding and catalysis. A similar trend was observed with temperature: activity increased from 25 °C to 35 °C, indicating that elevated kinetic energy enhances molecular collisions and reaction rates. However, activity declined at 45 °C, likely due to partial enzyme denaturation caused by heat disrupting the protein’s three-dimensional structure. These observations support the concept of an “optimum” temperature and pH for enzyme activity.
When compared to findings from other studies, these results are highly consistent. Previous research indicates that catalase typically has optimal activity around pH 7 and temperatures close to 37 °C, conditions that reflect the internal environment of most mammals and birds (Bartoszek, 2006; Robinson, 2015). Additionally, the observation that pig liver catalase consistently produced higher pressure than chicken liver catalase matches data from Gradinski-Vrbanac (2002), which reported that pig liver has catalase concentrations approximately 36 times higher than chicken liver. This substantial difference in enzyme quantity likely accounts for the increased reaction rate observed in the pig liver samples, as a higher enzyme concentration would result in more catalytic events occurring per unit time.
The underlying mechanisms that explain these trends are rooted in enzyme chemistry. Catalase, like all enzymes, relies on its active site structure to facilitate the breakdown of H₂O₂ into water and oxygen. At optimal pH, the active site maintains the correct charge and shape for effective binding and catalysis. Deviations from this pH disrupt the ionization of amino acids critical to the active site, reducing enzyme efficiency. Temperature plays a similar role—moderate heating increases kinetic energy and enhances enzyme-substrate interactions, while excessive heat destabilizes the hydrogen bonds and ionic interactions that maintain protein structure, leading to denaturation and reduced activity.
Despite the successful identification of these patterns, the study had several limitations. Some data at 45 °C had to be omitted due to experimental inconsistencies, such as possible temperature fluctuations or minor gas leakage, despite the use of a rubber stopper. Additionally, although efforts were made to keep liver mass and H₂O₂ concentration consistent, slight variations in liver homogenization or pipetting could have introduced small errors. Foam formation may have also trapped some of the generated oxygen, potentially reducing the recorded pressure and slightly underestimating enzyme activity. Increasing the number of replicates and improving the precision of sample preparation could help mitigate these limitations in future experiments.
This study serves to add understanding of how environmental conditions influence enzyme efficiency, which has potential implications for both biological systems and industrial applications. Understanding catalase’s optimal conditions can inform medical treatments for oxidative stress-related conditions, improve food processing protocols, and contribute to biotechnology advancements. Furthermore, by comparing catalase activity between species, this research delves into the physiological adaptations and antioxidant defenses that vary across animals.
Future studies could refine the temperature and pH ranges with smaller intervals—for example, testing every 2 °C between 30 and 40 °C or using finer pH increments near neutrality—to pinpoint exact optimal conditions. It would also be valuable to investigate catalase activity across different tissues within the same species to determine whether other organs possess more or less efficient catalase. Moreover, structural and genetic comparisons of pig and chicken catalase could provide insights into why pig liver catalase is significantly more effective, potentially identifying mutations or structural features responsible for increased activity.
References
Razaq, A. Haider, Z., Shahzad, M. T., Afzal, M. B. (2020) “Catalase Enzyme Role in Drug and Food Industry.” Global Scientific Journal, Global Scientific Journal. www.globalscientificjournal.com/researchpaper/Catalase_enzyme_indrug_and_food_industry.pdf
Checa, J., & Aran, J. M. (2020). Reactive Oxygen Species: Drivers of Physiological and Pathological Processes. Journal of inflammation research, 13, 1057–1073. https://doi.org/10.2147/JIR.S275595
Domínguez, L., Sosa-Peinado, A., & Hansberg, W. (2010). Catalase evolved to concentrate H2O2 at its active site. Archives of biochemistry and biophysics, 500(1), 82–91. https://doi.org/10.1016/j.abb.2010.05.017
Espinoza-Vergara, J., Molina, P., Walter, M., Gulppi, M., Vejar, N., Melo, F., Urzua, M., Muñoz, H., Zagal, J. H., Zhou, X., Azocar, M. I., & Paez, M. A. (2021). Effect of pH on the Electrochemical Behavior of Hydrogen Peroxide in the Presence of Pseudomonas aeruginosa. Frontiers in bioengineering and biotechnology, 9, 749057. https://doi.org/10.3389/fbioe.2021.749057
Gradinski-Vrbanac B, Stojevič Z, Milinkovič-Tur S, Balenovič T, Piršljin J, Zdeler-Tuk M. (2002) In vitro susceptibility of duck, chicken, and pig erythrocyte lipids to peroxidation. Vet Med-Czech. 47(10-11):303-308. https://doi.org/10.17221/5839-VETMED.
Heck, D. E., Shakarjian, M. Kim, H. D. Laskin, J. D., Vetrano, A. M. (2010) “Mechanisms of oxidant generation by catalase.” Annals of the New York Academy of Sciences. 120-5. https://doi.org/10.1111/j.1749-6632.2010.05603.x
Jomova, K., Raptova, R., Alomar, S. Y., Alwasel, S. H., Nepovimova, E., Kuca, K., & Valko, M. (2023). Reactive oxygen species, toxicity, oxidative stress, and antioxidants: chronic diseases and aging. Archives of toxicology, 97(10), 2499–2574. https://doi.org/10.1007/s00204-023-03562-9
Lismont, C., Revenco, I., Fransen, M. (2019). Peroxisomal Hydrogen Peroxide Metabolism and Signaling in Health and Disease. International journal of molecular sciences, 20(15), 3673. https://doi.org/10.3390/ijms20153673
Nandi, A., Yan, L. J., Jana, C. K., & Das, N. (2019). Role of Catalase in Oxidative Stress- and Age-Associated Degenerative Diseases. Oxidative medicine and cellular longevity, 2019, 9613090. https://doi.org/10.1155/2019/9613090
Ransy, C., Vaz, C., Lombes, A. Bouillaud, F. (2020) Use of H2O2 to Cause Oxidative Stress, the Catalase Issue. International journal of molecular sciences 21(23), 9149. 30. https://doi.org/10.3390/ijms21239149
Robinson P. K. (2015). Enzymes: principles and biotechnological applications. Essays in biochemistry, 59, 1–41. https://doi.org/10.1042/bse0590001
Tao, Z, Raffel, R.A, Souid, A.K., Goodisman, J., 2009. Kinetic studies on enzyme- cataluyzed reactions: oxidation of glucose, decomposition of hydrogen peroxide and their combination. Biophysical journal, 96(7), pg.2977-2988. https://doi.org/10.1016/j.bpj.2008.11.071
Tijskens, L. M., Greiner, R., Biekman, E. S., Konietzny, U. (2001) “Modeling the effect of temperature and pH on activity of enzymes: the case of phytases.” Biotechnology and bioengineering. 72(3).