Yanna Xiang – Applied Science, Year 2
Abstract
Steel is one of the foundational materials of modern infrastructure. Unfortunately, steels without additives are very vulnerable to corrosion, causing a decline in strength and reliability over time. Various corrosion prevention methods have been investigated and applied, with epoxy coatings and cathodic protection being the two most commonly used. While many other studies have examined the performance of either method against untreated steel, this experiment aimed to directly compare the efficacy of both epoxy coatings and cathodic protection against each other. This was done by repeatedly dipping steel samples with either treatment, as well as a control of untreated steel, into a 35ppt saline solution for 30 minutes at a time over the course of 16 days. It was found that both treatments were effective at preventing corrosion in comparison to the untreated control, although the cathodic protection was slightly less effective than the epoxy coating. Further research could incorporate a higher sample size and a longer time frame for more conclusive results.
Introduction
Steel is one of the most widely used materials in the built environment. It is both relatively inexpensive and quite strong, depending on its fabrication method. Various forms of steel are widely used in car manufacturing, as the framework holding up buildings, many infrastructure projects, pipes, tools, ships, and trains, among other things (Karimi, 2023). It is critical to the function of the modern world.
Given that there are so many different uses of steel as a material, naturally there are also a variety of ways to manufacture steel to manipulate its properties. Karimi (2023) explains that steel itself is an alloy of carbon and iron, which also may contain a selection of other metals, including but not limited to niobium, vanadium, titanium, chromium, and nickel. These other metals alter the properties of steel. Small amounts of either titanium, niobium, or vanadium (under 0.1%) can significantly strengthen the resulting steel alloy, while incorporating at least 10.5% chromium creates stainless steel which generally is highly resistant to corrosion (Karimi, 2023).
Despite the many beneficial properties that can be imparted onto steel through alloying, carbon steel – steel without additional metals or additives – is still the most widely used form of steel globally (Karimi, 2023). This is due to its relatively inexpensive price in comparison to other steels which require the inclusion of more expensive metals. This carbon steel can be further separated into high carbon steel and low carbon steel. Typically, the lower the carbon content, the more ductile and less brittle the steel is, while sacrificing its strength in return. Thus, for most construction and infrastructure projects, high carbon steel, also known as mild steel, is most widely used.
One of the challenges of mild steel though, is that without the inclusion of chromium, it is extremely vulnerable to corrosion, degrading its strength and reliability over time. This can occur in all manner of ways, including exposure to acidic, basic, and wet environments, as well as certain types of bacteria (Sarouhan et al., 1999). Because mild steel is used to provide strength to cars, buildings, and other infrastructure, if corrosion is left unchecked, it can often lead to very dangerous situations, like the recent collapse of the Fern Hollow Bridge in Pennsylvania (Hakimian, 2024).
Consequently, many methods have been developed to combat corrosion in mild steel, epoxy resin coatings and cathodic protection being the most common. According to a 2018 paper by Olayide Adetunji, epoxy resin coatings on mild steel form a barrier against exposure to environmental factors which may induce corrosion. Thus, these coatings are commonly used to protect steel in industrial environments, as well as in infrastructure (particularly projects that are public facing, as coatings can also provide decorative elements). In Adetunji’s study, epoxy coating was applied to mild steel samples which were then submerged alongside uncoated steel in either distilled water, 1M HCl solution, or 1M NaOH solution. It was found that the coating was extremely (95.3%) effective against corrosion in the distilled water when compared to the untreated samples, but only 49.6% effective in the HCl solution, and did not alter corrosion amounts in the NaOH. The study found that epoxy is best used in circumstances where the steel will be exposed to either water or acidic environments.
Cathodic protection is another method of corrosion prevention used to protect mild steels. One form of corrosion, galvanic corrosion, is an electrochemical process in which a current flows between a cathode and an anode, inducing corrosion in the anode, while delaying corrosion in the cathode. Under certain environmental conditions, like microbiologically induced damage, this can happen even within the same piece of metal (Sarouhan et al., 1999). Whichever metal, or section of metal, is more reactive, becomes the anode, while the less reactive part becomes the cathode. This can be utilized to protect mild steels by attaching a portion of a more reactive metal, like aluminum or zinc, to the surface of the steel according to a study by Johnson et al. (2019). This method is relatively easy to install and is quite low maintenance, although the anode is eventually used up and must be replaced. Another method of cathodic protection is coating the steel in a layer of the reactive metal as the steel is being manufactured, through a process known as galvanizing (Šmak et al., 2021). This is also very commonly used, although it is very difficult to apply to larger pieces of steel, as the steel needs to be submerged in the melted reactive metal. It can also be difficult to reapply after the anodic layer has worn off. Moreover, in the 2021 study conducted by Šmak et al., they found that the process of hot-dip galvanizing can severely damage the yield and tensile strengths as well as the hardness of steels due to the high temperatures involved in the process. Thus, for larger infrastructure projects, anode attachment is preferred if cathodic protection is used.
Direct comparisons between the performance of epoxy resin coatings and forms of cathodic protection have been done before, but not in the context of large infrastructure projects. Instead, like in Yeomans’s 1991 study, these comparisons have been addressed towards the efficacy of corrosion prevention within rebar for reinforced concrete. The study revealed that, while both epoxy resin coatings and zinc galvanization were effective at preventing corrosion of the steel bars, if the bars’ coatings were damaged in any way, the epoxy coated bars would perform significantly worse than the galvanized ones. This is because the salt water would be able to seep under the epoxy to corrode the steel while the zinc would continue acting as an anode regardless of damage to the coating. Although, once the zinc coating had been corroded through, the epoxy performed better as it was still able to protect portions of the rebar. The findings of this study, as they are based on galvanization, aren’t necessarily applicable to cathodic protection through anode attachment though.
Thus, there is an opportunity to add to the experimental knowledge regarding preservation of mild steel. Current studies either investigate the efficacy of individual methods of protection, like Olayide Adetunji’s 2018 research subjecting resin coatings to different conditions, or they compare epoxy and galvanic coatings in a different context, like that of construction and reinforced concrete (Yeomans, 1991). No paper has been found which compared the performance of cathodic protection through the attachment of anodes against an epoxy coating to find out which is more effective.
Materials and Methods
General
In order to test the efficacy of cathodic protection in comparison to an epoxy coating, an apparatus was constructed to raise and lower steel samples into and out of salt water at set intervals. The difference in weight of the samples would indicate the amount of corrosion which occurred over the course of the experiment.
Six rectangular samples, measuring 15.24 x 6.00 cm each, were cut from a 22 gauge sheet of steel. These were subdivided into pairs, each subject to a different treatment. Two samples received an epoxy coating, two were attached to an aluminum anode, and two were left untreated as controls.
The testing apparatus was constructed using a plastic bin to contain the saline solution, with wooden dowels spanning its length (Figure 1). The samples were placed in baskets and secured to the dowels with string. Through the rotation of the dowels, the string could either be wound up to raise the samples out of the saline, or released to submerge the samples. The dowel rotation was controlled by a continuous servo motor connected to the dowels by two gears, as the servo alone was unable to provide sufficient torque. All electrical components were secured to the lid of the tank, with the servo motor positioned at the edge, so that the gears could mesh together.
Figure 1: Apparatus at the start of testing as saline was poured in
Electronics
A FeeTech FS5103R continuous rotation servo motor was connected to a SparkFun RedBoard Plus using a solderless breadboard and wires. A wire was inserted into the servo motor’s black ground pin and wired to one of the blue (-) power rails of the breadboard. Another wire connected the end of the rail to one of the two ground pins on the RedBoard. The same was done with another set of wires, connecting the red power pin on the servo motor to the corresponding red (+) rail, which was then wired to the RedBoard’s 5V power supply pin. The white signal pin of the servo motor was finally then connected to pin (9) on the RedBoard, using a breadboard row to extend the wire.
The RedBoard was initially powered through its USB-C port, drawing power from a laptop while code was being written in the ArduinoIDE. The servo motor was then tested to find the “write()” value at which it would stop spinning. Next, code was drafted to allow the servo motor to spin clockwise for a second, stop for a second, spin counterclockwise for a second, stop for a second, before repeating itself. This confirmed the circuit was functional and the servo motor was performing as required.
Tank Set-up
For the experimentation vessel, two holes were drilled opposite each other, on the shorter faces of a 17” x 11 ⅛” x 12 ¾” plastic Sterilite storage bin, both 12 cm from the top. Four 12” wooden dowels were then hot glued together, with two end on end, and two others parallel on either side, centred around the joint. This created a flat middle section with two thinner, round dowel pieces protruding from either end. One end of the dowels was then cut so that they were short enough to fit into the storage bin, when being threaded through the holes.
As the servo motor would have been unable to lift the steel samples on its own, two gears of differing sizes were printed to allow for an increase of applied torque. The two gears were 3D printed using red PLA filament using a Bambu Studio X1 Carbon 3D printer. They were based on a resized .stl file from GregFrost’s “Parametric Involute Bevel and Spur Gears” post on Thingiverse. The plate sliced file settings were based on the Bambu Studio preset 0.20mm Standard @BBL X1C. The larger gear was hot glued onto the thin portion of the wooden dowels which stuck out of the plastic bin, so that the dowel acted as a shaft. A spare servo arm was then screwed on to the servo motor, before being epoxied to the centre of the smaller gear.
Six rectangular baskets with dimensions of 17 cm x 2 cm x 2.5 cm and holes in the top corners were also 3D printed – using red PLA on a Bambu Studio X1 Carbon 3D printer and the 0.20mm Standard @BBL X1C file slicing preset – in order to hold each of the steel samples.
Treatment Application
Once the sheet metal was cut, two pieces of steel were sanded using 200 grit sandpaper and cleaned using water, Dawn Platinum Dishwashing Liquid, and a sponge. Gorilla Epoxy was then applied on all sides and edges of both pieces, separately, using the included applicator. The samples were cured for a minimum of seven days before submersion (Figure 2).
Figure 2: The first epoxy sample before experimentation
A sheet of 0.019 inch thick aluminium was cut into eight 7.62 x 6.00 cm pieces. The aluminum anodes were secured to two steel samples using rubber bands. As each piece of aluminum was half the length and the same width as the steel samples, the samples were arranged with two pieces of aluminum, both in front and behind. In total, four aluminium pieces were attached to each sample of steel. Both samples were secured with a rubber band stretched lengthwise across the sample, and a rubber band looped diagonally widthwise around the middle (Figure 3).
Figure 3: The first aluminum sample before experimentation
Assembly and Experimentation
Each steel sample was weighed, before being placed in the 3D printed baskets. The baskets were placed into the plastic bin before string was threaded through the holes in the baskets and over the dowels above. The string was then evenly spaced and hot glued in place. Spacers of 3.5 cm were also cut from the remaining wooden dowels, and hot glued onto the sides of the baskets, so that they would remain consistently separated while being rotated in and out of the solution.
The servo motor was hot glued to the lid of the tank, arranged so that the teeth of its gear would mesh with the teeth of the gear on the dowel. The RedBoard was reprogrammed to move once every 30 minutes, to either reel up the steel samples into the air or to release them down into the saline solution. After the code had updated, the circuit was unplugged from the laptop and into a wall outlet to draw power for the duration of the trial.
A saline solution of 35 parts per thousand was then created using 280 grams of NaCl and 8 litres of water to mimic the salinity of seawater. The solution was poured into the plastic bin. The lid was put on and secured with masking tape. The trial ran for 16 days before the water was emptied (Figure 4). The samples were allowed to fully air dry before their masses were taken.
Figure 4. The experiment before water was emptied
Results
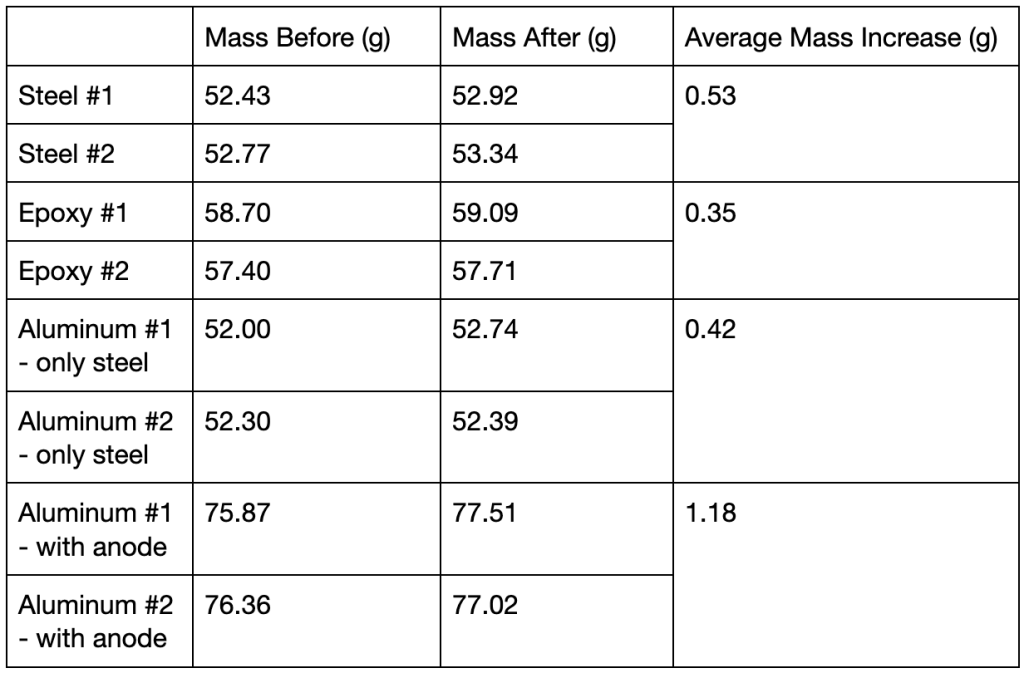
Mass measurements were taken both before and after exposure to saline solution (Table 1). The epoxy measurements were taken after the coating was applied. The steel samples were measured both with and without the aluminum anodes, but without the rubber bands in both cases.
Table 1: Measured masses of steel samples before and after testing
Discussion
Results
Throughout this experiment, the epoxy coated samples experienced the least amount of change in mass, while the control samples experienced the most. As seen in Table 1, on average the mass of the epoxy samples increased by 0.35g. The mass of the samples of steel treated with aluminum – weighed without the anodes – increased by 0.42g. The mass of the untreated samples increased by 0.53g. This indicates that both treatments did reduce the amount of corrosion, although the epoxy was more effective.
Errors and Improvements
A challenge faced by this project was the inconsistent angular velocity of the servo motor when moving clockwise as opposed to counterclockwise. As the servo motor doesn’t fully stop when programmed to 90 degrees, instead only stopping at approximately 93 degrees, its angular velocities when set to 0 and 180 are not equal in magnitude. Moreover, the difference was further increased as the servo spun faster when lowering the samples, as gravity increased its speed. This led to a gradual change in the start and end point of the steel baskets over time, as this discrepancy impact compounded. Thus, the number of rotations per second in each direction needed to be manually counted. With this information, the time the servo motor spent rotating in each direction was manually adjusted to account for the difference in angular velocity. This worked as the samples remained adequately elevated and submerged throughout the course of the experiment; should the experiment have run for longer, perhaps more issues would have emerged.
The application of the treatments could have also been a source of error. Should there have been small gaps left in the epoxy treatment, the steel would not have been fully coated and thus the saline solution would have been able to seep in and make contact with the sample. This is likely what happened with both epoxy samples. Furthermore, the preparation of the two epoxied samples may have differed from professional application. The sanding was intended to ensure a strong bond of the epoxy and metal, but any unevenness could have allowed the saline solution to seep between the two. This would have reduced the efficacy of the epoxy coating. The aluminum-anode protected samples were also subject to error due to the use of rubber bands to secure the aluminum and steel pieces together. If a rubber band was loose, less contact would occur between the two metals. This would reduce the efficacy of the cathodic protection and increase the amount of corrosion the steel underwent. This source of error would also account for the difference in corrosion between the two aluminium samples, as one set of rubber bands could have been tied tighter.
The experiment also only tested a limited number of samples for a short period of time. Thus, the results could differ from that of a large, long term infrastructure project that uses steel products.
Conclusion
Overall, it was found that both treatments reduced the amount of corrosion in the steel, with the aluminum anode coated samples averaging 0.07g more of a mass increase than the ones with epoxy coatings, indicating that slightly more corrosion had occurred. Thus, the epoxy coatings were found to be slightly more effective. Although the experiment was subject to large sources of error, when used in conjunction with other studies it can provide valuable insight into selecting the right corrosion prevention method for infrastructure construction materials. Future research could build on these results by using significantly larger sample sizes, maintaining the consistency of treatment application, and longer experimentation time.
References
Adetunji, O. (2018). Protective Efficiency of Epoxy Resin Coating on Mild Steel Plate in HCl, NaOH and Distilled Water. Umudike Journal of Engineering and Technology, 4(2). https://www.researchgate.net/publication/326837612_Protective_ Efficiency_of_Epoxy_Resin_Coating_on_Mild_Steel_Plate_in_HCl_NaOH_and_Distilled_Water
GregFrost. (2010, July 18). Parametric Involute Bevel and Spur Gears. Thingiverse. https://www.thingiverse.com/thing:3575.
Hakimian, R. (2024, March 22). Fern Hollow Bridge collapse: Details emerge on years of neglect leading to leg corrosion. New Civil Engineer. https://www.newcivilengineer.com/latest/fern-hollow-bridge-collapse-details-emerge-on-years-of-neglect-leading-to-leg-corrosion-23-02-2024/
Johnson, A. P., Lawler, J. S., & Murphy, M. S. (2019). Lessons in Galvanic Cathodic Protection Technology from Soldier Field and the Franklin Avenue Bridge. APT Bulletin: The Journal of Preservation Technology, 50. https://www.jstor.org/stable/26803331
Karimi, M. (2023). Review of Steel Material Engineering and Its Application in Industry. Journal of Engineering in Industrial Research, 4(1). https://doi.org/https://doi.org/10.48309/JEIRES.2023.1.6
Pietrowski, R., & Hiam, J. R. (1979). Fatigue Properties of Sheet Steel Before and After Galvanizing. SAE Technical Paper Series. https://doi.org/10.4271/790462
Sarouhan, B., Tedaldi, D., Lindsey, B., & Piszkin, A. (1999). Corrosion Woes. Water Environment & Technology, 11(4). https://www.jstor.org/stable/24667317
Šmak, M., Kubíček, J., Kala, J., Podaný, K., & Vaněrek, J. (2021). The Influence of Hot-Dip Galvanizing on the Mechanical Properties of High-Strength Steels. Materials, 14(18), 5219. https://doi.org/10.3390/ma14185219
Yeomans, S. R. (1991). Comparative Studies of Galvanized and Epoxy-Coated Steel Reinforcement in Concrete. “SP-126: Durability of Concrete: Second International Conference, Montreal, Canada 1991.” https://doi.org/10.14359/2214