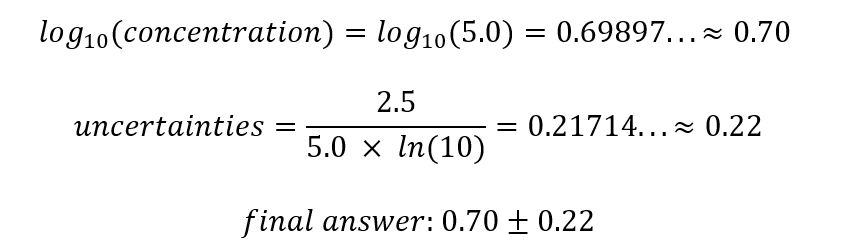Jamie Song – Life Science, Year 2
Abstract
Fermented foods like kimchi and sauerkraut have been long celebrated for their probiotic and nutritional benefits, yet concerns remain about the potential formation of harmful nitrites during fermentation. Nitrites can convert into carcinogenic nitrosamines under certain conditions, prompting questions about when fermented foods are safest to consume. This study investigates how varying the initial pH of fermentation solutions affects nitrite and nitrate levels over time, with the hypothesis that more acidic environments accelerate fermentation and suppress nitrite formation. Cabbage was fermented under three conditions: neutral with Kosher salt, acidic (10% acetic acid), and basic (10% sodium bicarbonate). Over 14 days, nitrate and nitrite levels were monitored using test strips, alongside pH readings. Results showed that the acidic condition rapidly dropped pH and maintained low nitrite levels, peaking at 0.75 ppm, while the basic condition delayed acidification and produced the highest nitrite accumulation (2.5 ppm). The salt-only group displayed moderate trends. These findings support the hypothesis and align with published literature (An et al., 2023), suggesting that pH manipulation is a practical strategy to improve fermentation safety. This work is particularly relevant to home fermenters and small-scale producers seeking low-tech methods to reduce health risks associated with nitrite buildup.
Introduction
The consumption of fermented foods has been a cultural and nutritional practice for thousands of years, valued for their flavor, preservation qualities, and health benefits. Fermented foods, including yogurt, kefir, sauerkraut, kimchi, and pickled vegetables, play a significant role in diets across cultures and are increasingly popular in contemporary health trends. This renewed interest was inspired by Sustainable Development Goal 3—to ensure healthy lives and promote well-being for all at all ages—by addressing concerns related to digestive health, immunity, and overall nutritional wellness.
One key benefit of consuming fermented foods stems from the presence of probiotics—live microorganisms that confer health benefits when consumed in adequate amounts. Probiotics support gut microbiota balance, promote digestion, and enhance immune function (Wang, X et al., 2021). During fermentation, microorganisms such as lactic acid bacteria, including Lactobacillus and Bifidobacterium,metabolize carbohydrates to produce bioactive peptides, short-chain fatty acids, and vitamins, enhancing the nutritional value of foods (Wang, Y et al., 2021).
However, despite the numerous health benefits, there is growing concern regarding the safety of consuming fermented foods at various stages of the fermentation process. Specifically, the accumulation of chemical compounds like nitrite—considered carcinogenic at high levels—has prompted questions about the health implications of fermented food consumption (Cantwell & Elliott, 2017). Understanding nitrite dynamics and identifying the optimal consumption timeline of fermented foods is thus crucial for maximizing health benefits while minimizing risks.
Current research has shown that fermentation helps enhance the metabolism activity, producing key bioactive compounds include biopeptides, vitamins, and conjugated linoleic acids, which are known for their protective effects on human health. CLAs, for instance, can help lower blood pressure and reduce inflammation by modulating oxidative stress pathways (Valenzuela et al., 2023). Studies on fermented dairy products such as yogurt and kefir demonstrate that probiotics can regulate the gut-brain axis, improve immune function, and alleviate symptoms of gastrointestinal disorders (Dahiya & Nigam, 2023).
In addition to CLAs, fermented vegetables contain compounds such as glucosinolates, which are metabolized into bioactive isothiocyanates during fermentation. These compounds exhibit anticancer properties and contribute to the nutritional enhancement of vegetables (Connolly et al., 2021). The production of such beneficial metabolites highlights the positive aspects of fermentation when carried out under controlled conditions.
Despite the significant focus on health-promoting metabolites, research has also indicated potential safety concerns. The accumulation of harmful byproducts, such as nitrite, can occur during the fermentation process, particularly in fermented vegetables. Nitrite formation is attributed to the activity of certain bacteria, which convert nitrates naturally present in vegetables into nitrites under anaerobic conditions (Tiso & Schechter, 2015). The presence of nitrite is concerning because it can react with secondary amines to form nitrosamines, chemical compounds classified as carcinogens due to their association with gastrointestinal cancers (Cantwell & Elliott, 2017).
The safety of fermented foods depends on factors such as the type of substrate, microbial composition, temperature, pH, and duration of fermentation. Studies have demonstrated that nitrite levels fluctuate during the fermentation timeline, often peaking at specific stages before declining as the process progresses (Lee & Yun, 2015). For instance, in the early stages of pickling vegetables, microbial activity leads to rapid nitrate reduction, resulting in increased nitrite concentrations. Research conducted on kimchi, a traditional Korean fermented vegetable dish, highlights this phenomenon. An et al. (2023) found that nitrite concentrations were highest within the first 3–7 days of fermentation at room temperature but declined as lactic acid bacteria (Lactobacillus plantarum) dominated the fermentation process. The acidification caused by the production of lactic acid suppresses the activity of nitrate-reducing bacteria, thereby reducing nitrite accumulation.
Despite these findings, there remains a lack of consensus regarding the safest stage to consume fermented foods, particularly for home-prepared products where fermentation conditions are often uncontrolled. Identifying the period during which nitrite levels are minimal is therefore essential for ensuring the safety of fermented foods and maximizing their health benefits. While existing research provides valuable insights into the health benefits and risks of consuming fermented foods, significant gaps remain. As most studies focus on the overall chemical composition of fermented foods without pinpointing the specific timeline during which nitrite levels are highest or lowest. Addressing these gaps is critical for providing evidence-based guidelines on the safest stages to consume fermented foods.
The present investigation aims to address these research gaps by systematically analyzing nitrite levels in fermented foods throughout the fermentation timeline. By identifying the stages at which nitrite levels are highest and lowest with varying pH levels, this study seeks to provide actionable recommendations for consumers and food producers to optimize the safety and health benefits of fermented foods.
Variables
The independent variable is the pH solution used to ferment the cabbage, manipulated by adding various amounts of acetic acid and sodium bicarbonate into the same volume solutions. The dependent variable is the nitrate and nitrite levels, which is determined by using test strips to approximate the values. The controls include the volume of solution to ferment the cabbage; the mass of cabbage added to each experimental solution; the time at which the nitrate and nitrite levels are measured; as well as the amount of sodium chloride added to each solution, in an attempt to mimic the fermentation method. This experiment assumes that the surrounding temperature will remain at SATP, Standard Ambient Temperature and Pressure, defined at 25°C and 100kPa, with negligible variation. We also assume that all reagents, unless stated otherwise, contains 100% purity.
Hypothesis
The pH of the solution used for natural fermentation of cabbage significantly affects the fermentation period, with more acidic conditions (lower pH) accelerating the fermentation process, while more alkaline conditions (higher pH) prolong it. As mentioned in both the background and by An et al. (2023), this effect is due to variations in microbial activity, where lower pH levels enhance lactic acid bacterial growth, thereby reducing nitrate and nitrite levels more rapidly.
Safety and Ethical Considerations
Ensuring the safety and ethics of this experiment is essential, given the involvement of chemical manipulation, microbial activity, and potential toxic byproducts. Proper handling of acetic acid and sodium bicarbonate is critical, as acetic acid is corrosive and can cause skin burns and eye irritation (PubChem, 2019), while sodium bicarbonate may irritate the respiratory tract if inhaled (PubChem, 2005). To prevent exposure, wear personal protective equipment (PPE) including lab gloves, goggles, and lab coats. Any chemical spills should be neutralized and cleaned immediately. Tools such as kitchen knives must be handled with care, avoiding contact with the blade and using them only under supervision.
Microbial growth presents another safety concern, as fermentation supports both beneficial and potentially harmful bacteria. Although lactic acid bacteria like Lactobacillus plantarum generally dominate (Cantwell & Elliott, 2017), contamination can occur under uncontrolled conditions. All equipment must be sterilized before and after use, and proper hand hygiene maintained. Fermented samples should be handled securely and never consumed, due to potential health risks. To minimize environmental impact, reusable materials such as glass jars should replace plastic. Experimental materials were scaled down by a factor of 10 to reduce waste while preserving integrity. Finally, all samples must be stored securely away from animals to avoid unintended ingestion of nitrite-containing or contaminated material.
Materials and Methodology
Preparing Reagents and Solutions
First, safety goggles, gloves, and lab apron were put on to ensure minimal exposure to hazardous reagents. Next, 450 grams of peeled cabbage was rinsed under tap water to remove dirt, residue, or rotten leaves. Each leaf was cut with a kitchen knife in approximate size of 2cm x 2cm. The chopped cabbage was then measured around 50g for each 260mL glass jar, and 5.00g of solid kosher salt was measured and dissolved—with a small spoon and an electric scale to two decimal places for precise measurements—in 50mL distilled water to receive a 10% brine solution. Two of such solutions were prepared in separate jars. After that, 3.00 g of solid sodium bicarbonate, NaHCO₃, was measured and dissolved in one of the 10% brine solutions, which was the experimental solution with higher pH (basic). Similarly, 5.00g of solid kosher salt was measured and dissolved in 50mL of 5% acetic acid, CH₃COOH, which was the experimental solution with lower pH (acidic). Thereafter, the digital pH meter was obtained and inserted into each solution, and corresponding values of initial pH were recorded in Table 1. The pH probe was ensured to be fully submerged in the solution for better consistency in the data recorded. The pH meter was also neutralized and cleaned before switching to measure another solution. Lastly, the above procedures were repeated whenever preparing solutions for a new testing trial.
Fermentation
Before handling any materials, hands were sterilized and protected with gloves. 50 grams of cleaned and cut cabbage was submerged into each glass jar, the cabbage was tightly packed with no visible air bubbles between the layers. This step helps create an anaerobic environment essential for proper fermentation. Next, the initial nitrite and nitrate levels were measured using test strips, carefully following the manufacturer’s instructions for accurate results. Measurements were recorded systematically. After documenting the data, each jar was securely sealed with its corresponding cap to prevent contamination. Jars were stored in a cool, dry environment away from direct sunlight. This storage condition was intended to control the experiment by minimizing external variables such as UV radiation that could affect the fermentation process. The fermentation was continuously monitored by remeasuring nitrite, nitrate, and pH levels every 24 hours from the start of natural fermentation. Consistent monitoring allowed for the observation of trends and changes over time. The entire process outlined above was repeated for each new trial, ensuring that a minimum of three replicates is performed for every test condition. After completing all trials, mean values of the collected data were taken to enhance the accuracy and reliability of the results.
Results
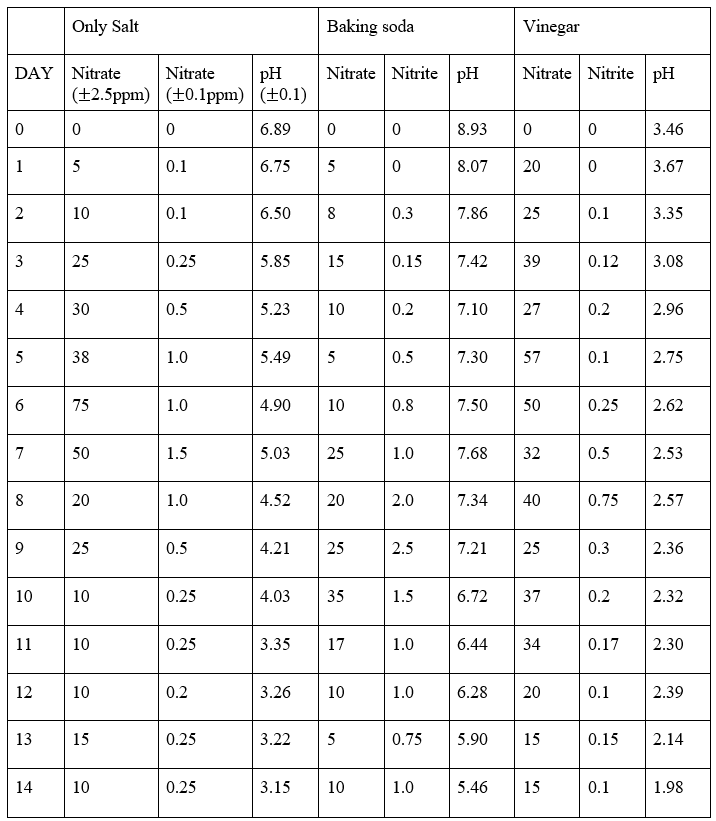
Appendix A displays all the calculated average of the three trials in which data is collected from this experiment. Sample calculation of the average procedure is shown below:
Sample Calculation Averaging all trials of measured Nitrate on Day 1 of the NaCl solution
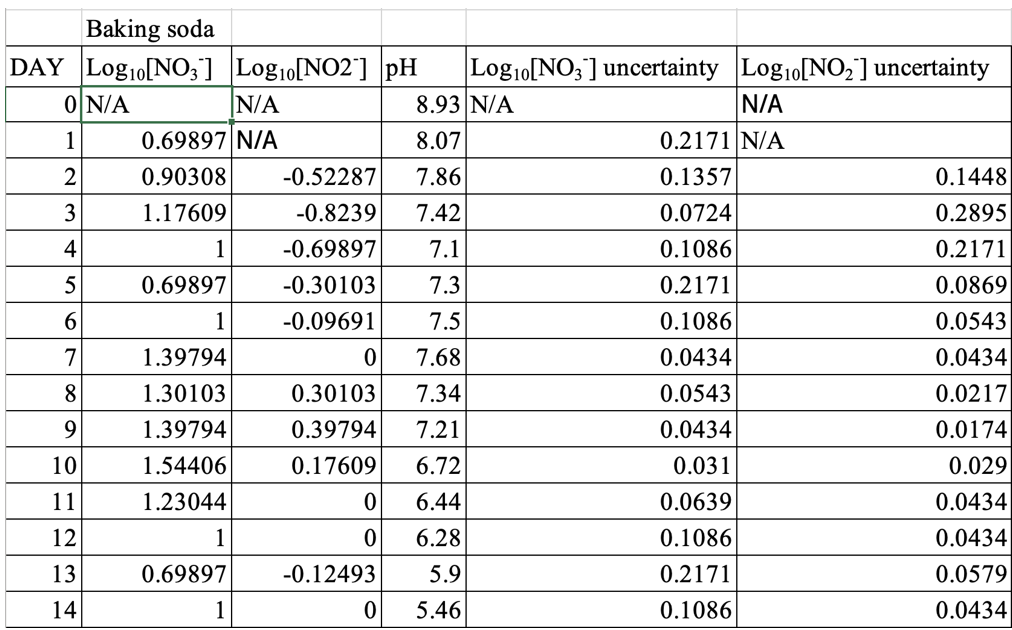
Due to the average nitrate levels being an order of magnitude above the average nitrite levels, the logarithms of the respective concentrations of the ions were graphed to allow easier comparisons. Steps to achieve the processed data is shown below:
Sample Calculation Finding the Logarithm of Nitrate of the Day 1, NaCl solution
The above calculation method were applied to process all other data collected, both nitrate and nitrite. However, the graphs with processed data excludes Day 0 measurements, since log10(0) is undefined. Refer to Appendix A for clarification. The net results for the processed data is displayed in Appendix B, Figures 1-5 show the graphs plotted according to calculated values:
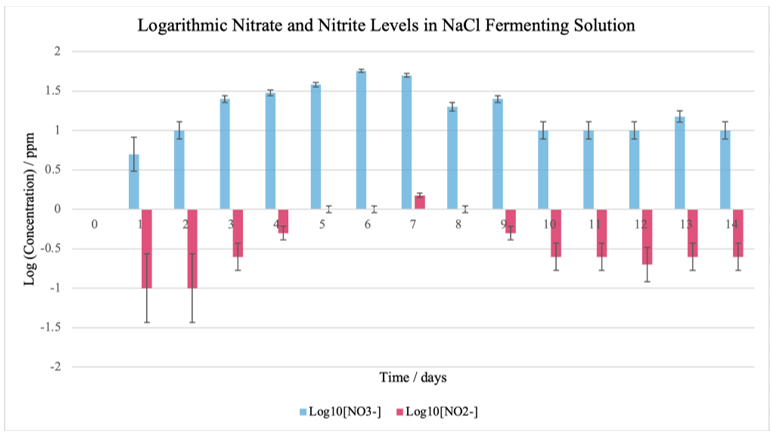
Figure 1: Graph comparing varying [NO3–] and [NO2–] in NaCl throughout the fermentation period
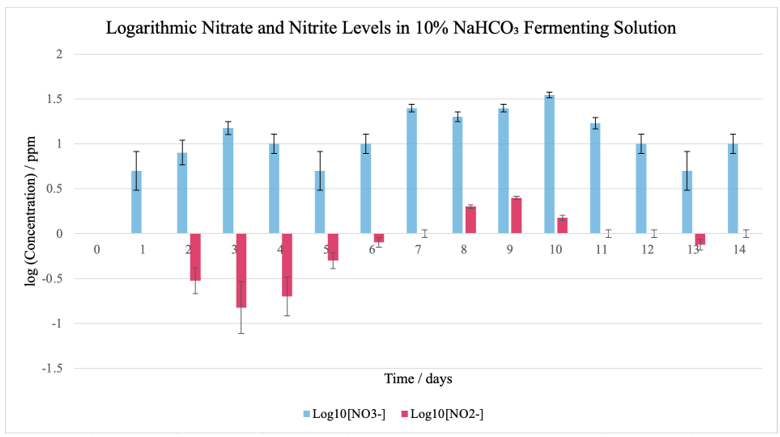
Figure 2: Graph comparing varying [NO3–] and [NO2–] in 10% NaHCO3 throughout the fermentation period
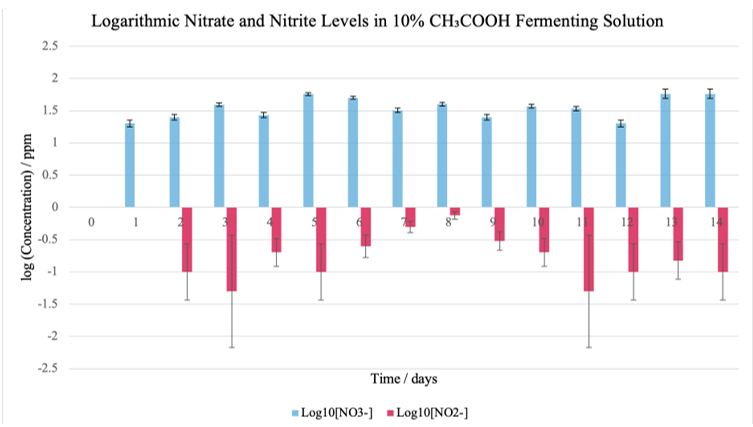
Figure 3: Graph comparing varying [NO3–] and [NO2–] in 10% CH3COOH throughout the fermentation period
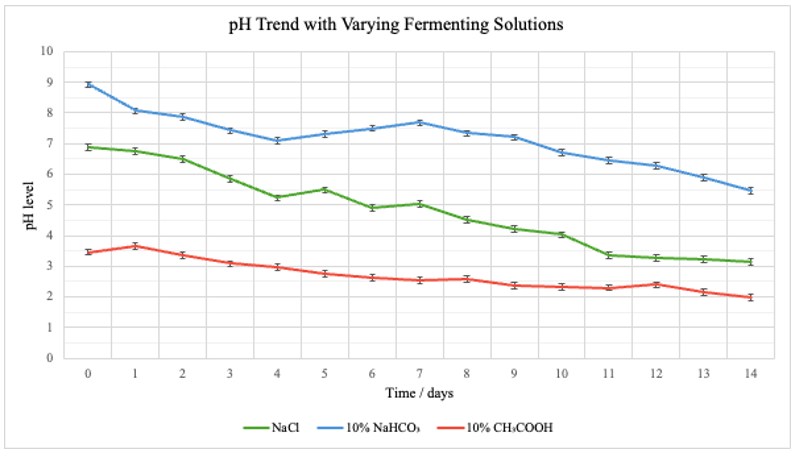
Figure 4: Graph comparing pH levels of the different solutions throughout the fermentation period
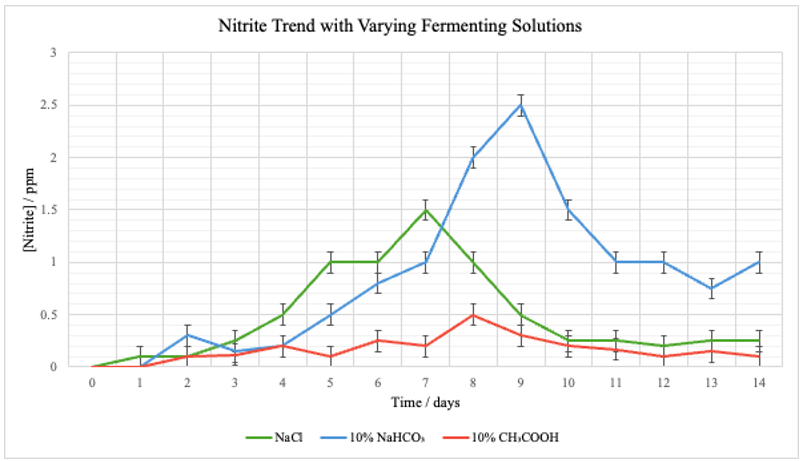
Figure 5: Graph comparing varying [NO2–] of different solutions throughout the fermentation period
Both Table 1 in Appendix A and Figures 1–5 demonstrate that the pH of the fermentation medium has a marked influence on both the chemical progression and safety indicators of the cabbage fermentation process. Across the three experimental conditions – salt (NaCl), baking soda (10% NaHCO3), and vinegar (10% CH3COOH) —distinct trends emerged in nitrate and nitrite levels as well as in the pH trajectories over the 14‐day fermentation period.
As seen on Figure 7, the pH for the NaCl group started at 6.89 and steadily declined to 3.15 by Day 14, suggesting a gradual acidification. Nitrate levels increased rapidly from 0 ppm at Day 0 to a maximum of 75 ppm on Day 6 before decreasing, while nitrite levels peaked at 1.5 ppm on Day 7.
Beginning with a higher initial pH of 8.93, exemplifying a more basic environment, the fermenting condition with 10% NaHCO3 exhibited a slower rate of acidification, reaching 5.46 by Day 14. Here, nitrate levels showed a more moderate rise—peaking at 35 ppm on Day 10—with nitrite reaching a higher maximum of 2.5 ppm on Day 9.
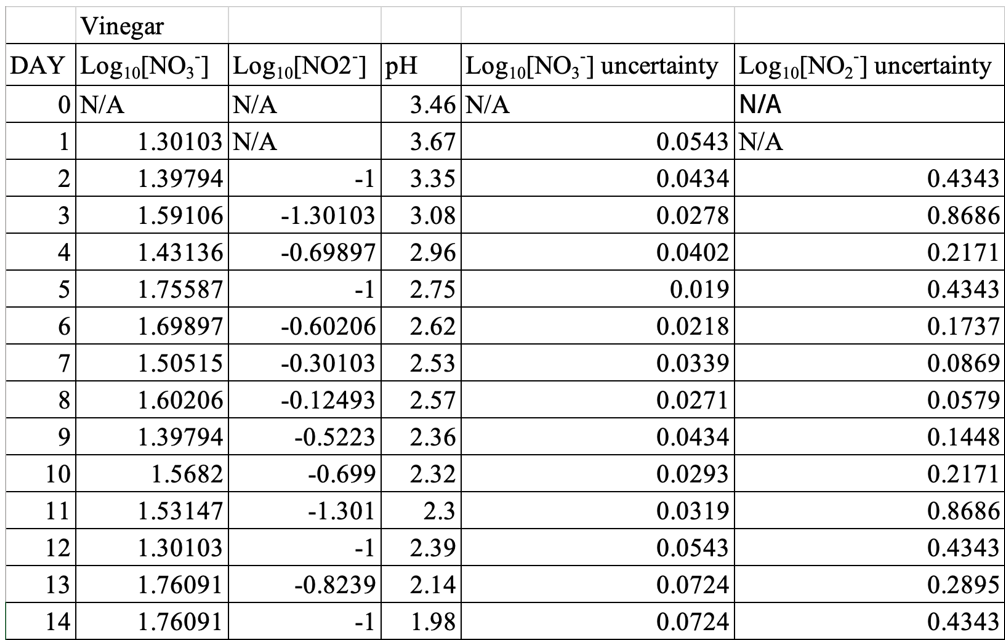
With an initial pH of 3.46, the experimental group with 10% CH3COOH maintained a consistently acidic environment (declining to 1.98 by Day 14). Although nitrate levels in this condition increased to a moderate peak (57 ppm on Day 5), nitrite levels remained relatively low—with a maximum of 0.75 ppm on Day 8—indicating that rapid acidification can effectively curb nitrite accumulation even when nitrate formation is evident.
Discussion
The investigation sought to determine how varying pH conditions during cabbage fermentation affect the period required for safe consumption in terms of fluctuating nitrite levels. The data collected in this experiment indicates that the initial pH of the fermentation medium may be a key determinant of both the rate of acidification and the nitrate and nitrite formation.
The data from the NaCl control group seen in Figure 7 indicates an early surge in microbial activity and chemical transformation—due to lactic acid bacteria producing lactic acid, thereby lowering the pH—that stabilizes as the environment gradually becomes more acidic. Compared to the control group, the basic fermenting environment with 10% NaHCO3 suggests that the delayed acidification may have allowed for extended nitrate-to-nitrite conversion, resulting in comparatively elevated nitrite accumulation during the mid-fermentation stages.
The experimental group with vinegar, 10% CH3COOH, had an inherently low initial pH, rapidly established an acidic environment that limited nitrite accumulation. In contrast, the experimental group with baking soda, 10% NaHCO3 had a higher starting pH and demonstrated a delayed acidification process and a higher peak in nitrite levels. These findings support the hypothesis that more acidic conditions accelerate fermentation and are associated with lower nitrite formation. The results are consistent with previous findings by An et al. (2023) and Lee and Yun (2015), where rapid acidification was observed to suppress the activity of nitrate-reducing bacteria, thereby reducing nitrite accumulation. Such comparisons reinforce the idea that pH control is critical in optimizing the safety of fermented foods.
The reduced nitrite levels in the vinegar group have significant implications on public health in consuming fermented foods, as lower nitrite formation can diminish the risk of nitrosamine production—a known carcinogenic pathway (Cantwell & Elliott, 2017). Although some nitrate differences in the later stages of fermentation did not achieve statistical significance, these non-significant differences still highlight important trends in microbial metabolism and chemical stability during fermentation.
A considerable strength of this experiment is its high ecological validity and its practical replicability. The experimental design closely mirrors traditional, real-world fermentation practices by employing common household reagents such as salt, baking soda, vinegar to modulate pH levels. This not only enhances the relevance of the findings for home-based and artisanal food production but also allows other researchers or practitioners to easily replicate and build upon the study.
However, this study is limited by the small sample size, with only three replicates per condition and three stages of variance, which restricts the statistical power of the findings. Additionally, utilizing test strips for nitrate and nitrite measurement introduces inherent uncertainties, as the colours on the strips are interpreted by the human eyes, which limits the precision of the data. Temperature fluctuation such as room drafts or nearby heat sources, may have been present during the fermentation stages, potentially affecting microbial activity. To address these limitations, future studies should include a larger number of replicates, employ more precise analytical methods, such as spectrophotometric assays, and ensure stricter environmental control, such as storing the fermenting vegetables in a closed system or space, to reduce variability.
The results of this investigation contribute valuable insights into the optimization of fermentation processes, especially in the context of home-based and artisanal food production. By establishing a rapid acidification, as seen in the acidic condition with 10% CH3COOH, lower nitrite accumulation can be achieved. These findings could have broader applications in improving the safety and nutritional value of fermented foods worldwide, while also providing a framework for further research into fermentation kinetics and microbial ecology.
In conclusion, the data support the hypothesis that lower initial pH conditions facilitate a safer fermentation process by expediting acidification and limiting nitrite accumulation. Although the results should be interpreted in light of the methodological limitations, they nonetheless provide a robust basis for recommending more acidic conditions in fermentation practices. This study underscores both the importance of pH control in food safety but also highlights avenues for further research in refining fermentation methods and enhancing public health outcomes.
References
An, J., Sun, L., Liu, M., Dai, R., Ge, G., Wang, Z., & Jia, Y. (2023). Influences of Growth Stage and Ensiling Time on Fermentation Characteristics, Nitrite, and Bacterial Communities during Ensiling of Alfalfa. Plants, 13(1), 84. https://doi.org/10.3390/plants13010084
Cantwell, M., & Elliott, C. (2017). Nitrates, Nitrites and Nitrosamines from Processed Meat Intake and ColorectalCancer Risk. Journal of Clinical Nutrition & Dietetics, 03(04). https://doi.org/10.4172/2472-1921.100062
Connolly, E. L., Sim, M., Travica, N., Marx, W., Beasy, G., Lynch, G. S., Bondonno, C. P., Lewis, J. R., Hodgson, J. M., & Blekkenhorst, L. C. (2021). Glucosinolates From Cruciferous Vegetables and Their Potential Role in Chronic Disease: Investigating the Preclinical and Clinical Evidence. Frontiers in Pharmacology, 12. https://doi.org/10.3389/fphar.2021.767975
Dahiya, D., & Nigam, P. S. (2023). Therapeutic and Dietary Support for Gastrointestinal Tract Using Kefir as a Nutraceutical Beverage: Dairy-Milk-Based or Plant-Sourced Kefir Probiotic Products for Vegan and Lactose-Intolerant Populations. Fermentation, 9(4), 388. https://doi.org/10.3390/fermentation9040388
Lee, H.-J., & Yun, Z.-S. (2015). Consumers’ perceptions of organic food attributes and cognitive and affective attitudes as determinants of their purchase intentions toward organic food. Food Quality and Preference, 39, 259–267. https://doi.org/10.1016/j.foodqual.2014.06.002
PubChem. (2005, July 19). Sodium carbonate. Nih.gov; PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/Sodium-carbonate
PubChem. (2019). Acetic acid. Nih.gov; PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/Acetic-acid
Tiso, M., & Schechter, A. N. (2015). Nitrate Reduction to Nitrite, Nitric Oxide and Ammonia by Gut Bacteria under Physiological Conditions. PLOS ONE, 10(3), e0119712. https://doi.org/10.1371/journal.pone.0119712
Valenzuela, C. A., Baker, E. J., Miles, E. A., & Calder, P. C. (2023). Conjugated Linoleic Acids Have Anti-Inflammatory Effects in Cultured Endothelial Cells. International Journal of Molecular Sciences, 24(1), 874. https://doi.org/10.3390/ijms24010874
Wang, X., Zhang, P., & Zhang, X. (2021). Probiotics Regulate Gut Microbiota: An Effective Method to Improve Immunity. Molecules, 26(19), 6076. https://doi.org/10.3390/molecules26196076
Wang, Y., Wu, J., Lv, M., Shao, Z., Hungwe, M., Wang, J., Bai, X., Xie, J., Wang, Y., & Geng, W. (2021). Metabolism Characteristics of Lactic Acid Bacteria and the Expanding Applications in Food Industry. Frontiers in Bioengineering and Biotechnology, 9(612285). https://doi.org/10.3389/fbioe.2021.612285
Appendix A
Table 1: Averaged Values of Nitrate, Nitrite, and pH Change Throughout the Experiment
Appendix B
Table 1: Logarithmic Averaged Values of Nitrate, Nitrite, with uncertainties, and pH Change Throughout the Experiment, for Only Salt
Table 2: Logarithmic Averaged Values of Nitrate, Nitrite, with uncertainties, and pH Change Throughout the Experiment, for Sodium Bicarbonate
Table 3: Logarithmic Averaged Values of Nitrate, Nitrite, with uncertainties, and pH Change Throughout the Experiment, for Acetic Acid