Ella Xie – Life Science
Abstract
The purpose of this experiment was to distinguish the relationship between soil acidity and degradation of a plant’s rhizosphere. The concentration of gram-negative bacteria was observed as soil bacteria in the rhizosphere is mainly gram-negative. Individual containers of broccoli sprouts Brassica oleracea were exposed to different pH levels of lemon juice water mixtures, representing acid rain. Suspension sampling was used to prepare soil samples for plating on Petri dishes. Two samples of each pH were taken before and after acidification. Each sample was then Gram-stained to compare the amount of Gram-negative bacteria present before and after acidification. Broccoli sprouts exposed to a lower pH level were found to have less visible bacterial growth present on the roots than those of a higher pH level. The plants that had more bacterial growth appeared to be in worse condition, but Gram-staining revealed no significant difference between different pH levels. Further research with proper enumeration and bacterial determination procedures is needed to determine the effect of acidification on plant rhizospheres.
Introduction
Human industries occupy one-fifth of the land mass on earth. Ecosystems that are influenced by humans due to industrial expansion gradually become barren due to overuse, depleting the land of essential natural resources such as vegetation and clean water (Paxman, 2020).
The environment is industrialized to accommodate the high demand for resources generated by the human populace at the detriment of natural cycles responsible for maintaining homeostasis within ecosystems (EPA, 2020). The nitrogen cycle regulates the exchange and release of nitrogen between the atmosphere and surrounding ecosystems (Bernhard, 2010). The rapid increase in agricultural production and deforestation from the industrial age causes an imbalance in nitrogen cycling, with extended agricultural greenhouse gas (GHG) emissions unable to be managed by depleted forests (Vitousek, 1981). The unmanaged nitrogen is then released into the atmosphere and combines with its abundant water molecules, forming acid rain (New Hampshire, 2019), causing the over-acidification of once-fertile farmland observed all over the world (Tian & Niu, 2015).
Soil acidification is the build up of acidity in soil over time, caused by the overuse of nitrogen-based fertilizers, acid rain, and nitrogen cycle leeching (Queensland, 2013). It contributes to an overall decrease in plant health, depriving plants of their essential biological functions and nutrients (Agriculture Victoria, 2020).
Soil acidification significantly harms plant health, since it drains the plant rhizosphere of helpful bacteria and essential nutrients. A plant’s rhizosphere is the soil that influences the maintenance of the roots that ensure the plant’s growth. Gram-negative bacteria tend to be acidophilic; meaning that it thrives in high-acid environments (Johnson, 1998). Therefore, this experiment’s hypothesis outlines that decreasing the soil pH of the broccoli sprouts (Brassica oleracea) through consistent watering using lemon water (pH 4, 5, 6, and 7) will have an inversely proportional relationship with the concentration of Gram-negative bacteria present within the rhizosphere. Alternatively, the null hypothesis occurs when increasing the soil pH of the broccoli sprouts through consistent watering does not influence the concentration of gram-negative bacteria present within the rhizosphere.
Materials and Methodology
The broccoli sprout seeds (Mumm’s) were grown in soil and potted in clear plastic jars. Twelve samples of broccoli sprouts were made. Sprouts were then regularly watered with around 15mL of pH 7 tap water bidaily for four days. A soil sample was taken from each plant. Rhizospheric bacteria was isolated using suspension dilution of the soil at a 1:1000 part dilution ratio in order to isolate bacterial colonies and prevent lawn growth on Petri dishes. Nutrient agar dishes were prepared for plating. Tealight candles were lit to create an updraft to prevent contamination. Solution I was plated onto the nutrient agar plates using a flame-sterilized L-spreader. The plates were left for a 1-week incubation period. The bacterial growth on the plates was then observed and recorded corresponding with each soil sample.
On the fifth day, the sprouts were separated into four distinct groups (pH 4, pH 5, pH 6, pH 7; with pH 7 being the control samples) to receive different pHs of a water-lemon juice mixture used to model acid rain. The plants were then left untouched for a two-day growth period to allow the plant’s rhizosphere to absorb the lemon water. Subsequently, a soil sample was taken from each plant. The soil samples were then diluted and plated on petri dishes with the aforementioned plating procedure. The petri dishes were then left to grow for one week. Bacteria was then collected from the petri dishes and heat fixed onto microscope slides for Gram staining. The microscope slides were then gram-stained using Crystal Violet, Gram’s Iodine (2 parts Potassium Iodine, 1 part liquid Iodine solution in 300mL of water), 95% Ethanol, and Safranin. Gram-negative bacteria were enumerated and observed between the different pH levels of soil sampled before the acid exposure and after the acid exposure.
Results
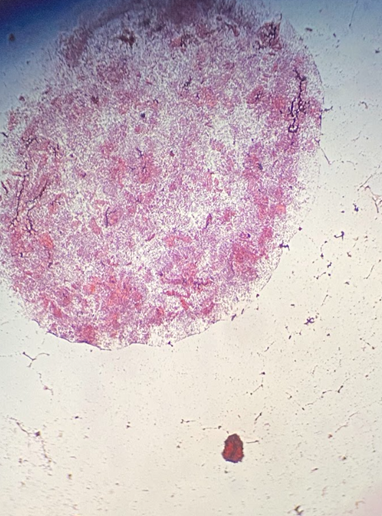
The concentration of Gram-negative bacteria was analyzed based on the factors of visible bacterial growth, root length, and enumeration through Gram-staining. Although the Gram-staining procedure provided no enumerated difference between pH levels (Figure 1, Figure 2), the broccoli sprouts still had visible differences in their growth. The sprouts grown at a consistent pH level of 7 were short, messy, and had wrinkled yellow leaves (Figure 3). Blue-toned grey fungal growth is scattered and interspersed within the root system (Figure 4). Contrarily, the sprouts grown at a consistent pH level of 4 retained an upright and straight growth direction with faded green leaves. The root system seems fully intact and relatively absent from any bacterial overgrowth (Figure 5).
Figure 1- Gram-stained sample of pH 4 soil
Figure 2- Gram-stained sample of pH 7 soil
Figure 3- Root system of pH 7 sample
Figure 4- Visible bacterial growth on pH 7 root system
Figure 5- Root system of pH 4 sample
Discussion
The purpose of this research study was to evaluate the effect of acidification on the presence of gram-negative rhizospheric bacteria. The visible bacterial growth on the plants suggest that lowering the pH of the water used for a plant decreases the amount of bacterial growth within a plant’s rhizosphere, rejecting the initial hypothesis. However, as the gram-staining procedure was unable to discern a difference between the concentration of Gram-negative bacteria present at different pH levels, further research is required to consolidate this claim. An inversely proportional relationship between the pH of an environment and bacteria growth does not align with previous knowledge of Gram-negative bacteria in acidified environments, as Gram-negative bacteria tend to be acidophilic and thrive in acidified (low-pH) environments (Johnson, 1998).
The results of this experiment are to be interpreted only within the constraints of the outlined procedure. This experiment was conducted within a lab environment, meaning that it may not have high ecological validity; It is limited by factors that cannot be found without the presence of an open system. Furthermore, the modelled acid rain can only be used to gauge a plant’s response to constant acidification from different pH levels, negating the impact of the chemical composition of acid rain on plants (Koch, 2023). B. oleracea sprouts reach maturation swiftly and have short growing seasons, meaning that the rhizospheres might not have enough time to produce a discernible difference in bacterial concentration before sampling. Due to that factor, this study also may not be applicable to other plant species.
A major limitation on this research study was the apparatus used to quantify bacteria. Soil contains over 60 thousand species of bacteria (Reid & Wong, 2005), and Petri dishes can only hold around 30-300 colonies of bacteria before growth cannot be enumerated (Toole, 2016). Although the soil samples were diluted down to 4.24 x 10 -2 percent of the samples’ original concentrations, some of the plates still grew bacterial lawns. This could have made the microscope slides indistinguishable in bacterial concentration. Therefore, the Gram-staining procedure was unable to present a noticeable difference between pH samples.
Overall, the qualitative results observed seem to show a positive correlation between pH level of soil and bacterial growth. As the bacteria could not be effectively enumerated, a stronger research claim with more effective bacterial enumeration and classification procedures should be carried out to verify the findings of this study, as soil acidification has become an increasingly relevant problem with the onslaught of climate change.
References
Bernhard, A. (2010). The Nitrogen Cycle: Processes, Players, and Human Impact. Nature Education Knowledge. https://www.nature.com/scitable/knowledge/library/the-nitrogen-cycle-processes-players-and-human-15644632/
Department of Jobs, P. and R. (2020, December 29). Soil acidity – Agriculture. Agriculture Victoria. https://agriculture.vic.gov.au/farm-management/soil/soil-acidity#:~:text=decrease%20the%20availability%20of%20essential
Environment and Climate Change Canada. (2023). GREENHOUSE GAS EMISSIONS CANADIAN ENVIRONMENTAL SUSTAINABILITY INDICATORS. https://www.canada.ca/content/dam/eccc/documents/pdf/cesindicators/ghg-emissions/2023/greenhouse-gas-emissions-en.pdf
Johnson, D. B. (1998). Biodiversity and ecology of acidophilic microorganisms. FEMS Microbiology Ecology, 27(4), 307–317. https://doi.org/10.1111/j.1574-6941.1998.tb00547.x
Koch Agronomic Services. (2023, December 21). The Role of Nitrogen in Crop Production and How To Protect It. Kochagronomicservices.com. https://kochagronomicservices.com/knowledge-center/The-Role-of-Nitrogen-in-Crop-Production-and-How-to-Protect-It
New Hampshire Department of Environmental Services. (2019). Acid Rain. In New Hampshire Department of Environmental Services. https://www.des.nh.gov/sites/g/files/ehbemt341/files/documents/2020-01/bb-8.pdf
O’Toole, G. A. (2016). Classic Spotlight: Plate Counting You Can Count On. Journal of Bacteriology, 198(23), 3127–3127. https://doi.org/10.1128/jb.00711-16
Paxman, G. (2020, August 25). 50% of Earth’s Land Surface Remains Relatively Untouched By Humans. Earth.org. https://earth.org/half-of-earths-land-surface-remains-relatively-untouched-by-humans/#:~:text=Currently%2020%25%20of%20Earth
Queensland Government. (2013). Soil acidification. Qld.gov.au. https://www.qld.gov.au/environment/land/management/soil/soil-health/acidification
Reid, G., & Wong, P. (2005). Soil bacteria. https://www.dpi.nsw.gov.au/__data/assets/pdf_file/0017/41642/Soil_bacteria.pdf
Ritchie, H., Rosado, P., & Roser, M. (2020). Greenhouse gas emissions. Our World in Data. University of Oxford. https://ourworldindata.org/greenhouse-gas-emissions#:~:text=The%20average%20
Tian, D., & Niu, S. (2015). A global analysis of soil acidification caused by nitrogen addition. Environmental Research Letters, 10(2), 024019. https://doi.org/10.1088/1748-9326/10/2/024019
US EPA. (2021, April 15). Causes of Climate Change. http://www.epa.gov. https://www.epa.gov/climatechange-science/causes-climate-change#:~:text=Since%20the%20Industrial%20Revolution%2C%20human
Vitousek, P. M. (1981). Clear-Cutting and the Nitrogen Cycle. Ecological Bulletins, 33, 631–642. JSTOR. https://www.jstor.org/stable/45128696